PHYSIOLOGY | IMAGES | ETYMOLOGY | TAXONOMY | GEOGRAPHY | BEHAVIOR | DIET | REPRODUCTION | ECOLOGY | CONSERVATION | FAUNAFACTS | VIDEO | SOURCES

The hawksbill turtle is a sea turtle that can be found in shallow, warm waters off the coast of nearly every continent, aside from Antarctica. Named for the sharp, pointed beak that resembles the beak of a bird of prey, this turtle can be easily identified from others, such as the green turtle. A migratory, species, the hawksbill travels miles within the seas, but always returns to its place of birth to breed and lay its own eggs. Unfortunately, the trip from the beach to the ocean is a dangerous adventure for newborn hawksbill turtles.
Physiology
The hawksbill turtle’s appearance is similar to that of other marine turtles.
In general, the hawksbill turtle has a flattened body shape, flipper-like limbs adapted for swimming in the open ocean, and a protective carapace.
Hawksbill turtles, like all turtles, have a hard shell that discourages predators from trying to eat them. The beautiful carapace of the hawksbill turtle is generally streaked and marbled with amber, yellow, or brown, and can slightly change colors, depending on water temperature. Young hawksbill turtles have a heart-shaped carapace. As these turtles mature, their carapaces become more elongated.
With the exception of very old individuals, hawksbill turtles often have a strongly serrated edge on the lateral and posterior areas of their shells, giving a saw-like appearance of the shell margins.
Unlike other marine turtles, the thick scales, or scutes, of the hawksbill turtle’s carapace are imbricate, or overlapping, hence the scientific name, imbricata. These scales have four pairs of costal scutes.
The heads of hawksbill turtles have two pairs of prefrontal scales.
The elongated mouth of the hawksbill turtle resembles a beak that tapers off to a sharp point at the end. The sharp, curving beak has prominent tomium, the sharp cutting edge.
Hawksbill turtles have two claws on each of their forelimbs.
There is sexual dimorphism in the physiology of the hawksbill turtle. Male hawksbill turtles are distinguished from females by a brighter pigmentation, a concave plastron, long claws, and a thicker tail.
Hawksbill turtles are relatively small sea turtles. They range from 76-89 centimeters, (30-35 inches,) in length and 40-68 kilograms, (90-150 pounds,) in weight. Nesting females average a length of 87 centimeters in curved carapace length and weigh up to 80 kilograms. The average hatchling in the parts of the Caribbean, owned by the United States, is about 42 millimeters in straight carapace length and weighs 13.5 to 19.5 grams.
Hawksbill turtles are long-lived and mature very slowly, taking 20 to 40 years to fully develop. The normal lifespan of hawksbill turtles is thought to be about 20 to 50 years, however biologists are not sure exactly how long they live. In the Caribbean and Western Atlantic, hawksbills may mature in 20 or more years. Age to maturity in the Indo-Pacific, however, requires a minimum of 30-35 years.
Generation length is defined as the age to maturity plus one half the reproductive longevity. Given estimated ages to maturity of hawksbill turtles at 25 years in the Caribbean and 35 years in the Indo-Pacific, with half of reproductive longevity estimated at 10 years, a conservative generation length of 35 years, (25 + 10 years,) is calculated for the Caribbean and Western Atlantic, and 45 years, (35 + 10 years,) in the IndoPacific. In analyzing the data, declines over three generations are therefore measured for up to 105 years in the Caribbean and Western Atlantic and up to 135 years in the Indo-Pacific. In fact, generation length may well have been longer in the days when population density was higher.

SEXUAL DIMORPHISM
Brighter Males with Concave Plastrons, Long Claws, & Thicker TailsBODY LENGTH
65-89 cm. / 25-35 in.BODY MASS
40-80 kg. / 90-177 lb.LIFESPAN
20-50 yr.GENERATION LENGTH
35-45 yr.

Images
Taxonomy
The hawksbill turtle is one of the seven extant species of sea turtles: green turtle (Chelonia mydas), loggerhead turtle (Caretta caretta), Kemp’s ridley (Lepidochelys kempii), olive ridley (Lepidochelys olivacea), flatback (Natator depressus), and leatherback (Dermochelys coriacea).
Hawksbill turtles have five features that distinguish them from other sea turtles. The heads of hawksbill turtles have two pairs of prefrontal scales, compared to the one pair of prefrontal scales of the green turtle. Hawksbill turtles also have two claws on each of their forelimbs and thick, overlapping scutes on their carapaces, which also have four pairs of costal scutes.
The hawksbill turtle is easily distinguished from other sea turtles by its sharp, curving beak with prominent tomium, the sharp cutting edge. The elongated mouth of the hawksbill turtle resembles a beak that tapers off to a sharp point at the end. This contrasts with the short, rounded beak of the green turtle.
Genetic analyses in the Atlantic and Indo-Pacific indicate that nesting populations comprise separate and identifiable stocks that should be treated as separate management units. Hawksbill aggregations on foraging grounds comprise animals from multiple nesting populations and often include animals from distant rookeries.
KINGDOM
AnimaliaPHYLUM
ChordataCLASS
ReptiliaORDER
TestudinesFAMILY
CheloniidaeGENUS
EretmochelysSPECIES
imbricataSUBSPECIES (3)
E. i. imbricata (Atlantic), E. i. bissa (Indo-Pacific, Pacific), E. i. squamata (Eastern Pacific)
Etymology
The elongated mouth of the hawksbill turtle resembles a beak that tapers off to a sharp point at the end. This strongly hooked, elongated V-shaped tapered point gives the turtle its common name, hawksbill turtle, as its beak resembles that of a bird of prey.
Unlike other marine turtles, the thick scales, or scutes, of the hawksbill turtle’s carapace are imbricate, or overlapping, hence the scientific name, imbricata.
GROUP
Bale, Dole, Nest, TurnYOUNG
Hatchling
Region
The hawksbill turtle has a wide, circumglobal distribution throughout tropical and, to a lesser extent, subtropical waters of the Atlantic Ocean, Indian Ocean, and Pacific Ocean.
Hawksbill nesting occurs in at least 70 countries, although much of it now only at low densities. Their movements within the marine environment are less understood, but hawksbills are believed to inhabit coastal waters in more than 108 countries.
In the Atlantic, hawksbill populations range as far west as the Gulf of Mexico and as far southeast as the Cape of Good Hope in South Africa. They live off the Brazilian coast, specifically Bahia, Fernando de Noronha, through southern Florida and Virginia waters.
In the Caribbean, the main nesting beaches are in the Lesser Antilles, Barbados, Guadeloupe, Tortuguero in Costa Rica, and the Yucatan. They feed in the waters off Cuba and around Mona Island near Puerto Rico, among other places.
In the western hemisphere, they have been reported to have nests as far north as Woods Hole, Massachusetts. They are also present in the Long Island Sound. However, between the Carolinas and New Jersey, very few hawksbill turtles have been recorded.
In the Indian Ocean, hawksbills are a common sight along the east coast of Africa, including the seas surrounding Madagascar and nearby island groups. Hawksbills are also common along the southern Asian coast, including the Persian Gulf, the Red Sea, and the Indian subcontinent and Southeast Asia coasts. They are present across the Malay Archipelago and northern Australia. Their Pacific range is limited to the ocean’s tropical and subtropical regions. In the west, it extends from the southwestern tips of the Korean Peninsula and the Japanese Archipelago south to northern New Zealand.
The Philippines hosts several nesting sites, including the island of Boracay and Punta Dumalag in Davao City. Dahican Beach in Mati City, Davao Oriental, hosts one of the essential hatcheries of its kind, along with olive ridley sea turtles in the archipelagic country of the Philippines. A small group of islands in the southwest of the archipelago is named the “Turtle Islands” because two species of sea turtles nest there: the hawksbill and the green sea turtle. In January 2016, a juvenile was seen in Gulf of Thailand. A 2018 article by The Straits Times reported that around 120 hawksbill juvenile turtles recently hatched at Pulau Satumu, Singapore. Commonly found in Singapore waters, hawksbill turtles have returned to areas such East Coast Park and Palau Satumu to nest. In Hawaii, hawksbills mostly nest on the “main” islands of Oahu, Maui, Molokai, and Hawaii. In Australia, hawksbills are known to nest on Milman Island in the Great Barrier Reef. Hawksbill sea turtles nest as far west as Cousine Island in the Seychelles, where the species since 1994 is legally protected, and the population is showing some recovery. The Seychelles’ inner islands and islets, such as Aldabra, are popular feeding grounds for immature hawksbills.
In the eastern Pacific, hawksbills are known to occur from the Baja Peninsula in Mexico south along the coast to southern Peru. Nonetheless, as recently as 2007, the species had been considered extirpated mainly in the region. Important remnant nesting and foraging sites have since been discovered in Mexico, El Salvador, Nicaragua, and Ecuador, providing new research and conservation opportunities. In contrast to their traditional roles in other parts of the world, where hawksbills primarily inhabit coral reefs and rocky substrate areas, in the eastern Pacific, hawksbills tend to forage and nest principally in mangrove estuaries, such as those present in the Bahia de Jiquilisco (El Salvador), Gulf of Fonseca (Nicaragua, El Salvador, and Honduras), Estero Padre Ramos (Nicaragua), and the Gulf of Guayaquil (Ecuador). Multi-national initiatives, such as the Eastern Pacific Hawksbill Initiative, are currently pushing efforts to research and conserve the population, which remains poorly understood.
EXTANT
American Samoa, Antigua & Barbuda, Australia, Bahamas, Bahrain, Barbados, Belize, Bonaire, Sint Eustatius & Saba, Brazil, British Indian Ocean Territory, Cambodia, China, Colombia, Comoros, Costa Rica, Cuba, Curaçao, Dominican Republic, Ecuador, Egypt, El Salvador, Equatorial Guinea, Eritrea, Fiji, French Southern Territories, Grenada, Guadeloupe, Guam, Honduras, India, Indonesia, Iran, Jamaica, Japan, Kenya, Kuwait, Madagascar, Malaysia, Maldives, Martinique, Mauritius, Mayotte, Mexico, Micronesia, Mozambique, Myanmar, Nicaragua, Oman, Palau, Panama, Papua New Guinea, Philippines, Puerto Rico, Qatar, Saint Kitts & Nevis, Saint Martin, Samoa, Sao Tome & Principe, Saudi Arabia, Seychelles, Sint Maarten, Solomon Islands, Somalia, Sri Lanka, Sudan, Taiwan, Tanzania, Thailand, Trinidad & Tobago, United Arab Emirates, United States, Vanuatu, Venezuela, Vietnam, Virgin Islands, YemenUNCERTAIN
Cayman Islands, Sierra Leone
Habitat
Hawksbill turtles are most commonly found in marine neritic, oceanic, and intertidal developmental foraging habitats that may comprise coral reefs or other hard-bottom habitats containing sponges.
They also reside in shoals, sea grass, algal beds, mangrove bays, lagoons of oceanic islands, creeks, mud flats, and continental shelves.
As they increase in size, immature hawksbill turtles typically inhabit a series of developmental habitats, with some tendency for larger turtles to inhabit deeper sites.
In general, hawksbill turtles are found in water no deeper than sixty feet, or 18.3 meters.
Of all the sea turtle species, the hawksbill turtle is the one most associated with warm, tropical waters.
MARINE NERITIC
Pelagic, Subtidal Rock & Rocky Reefs, Subtidal Muddy, Macroalgal/Kelp, Coral, Submerged SeagrassMARINE OCEANIC
EpipelagicMARINE INTERTIDAL
Sandy Shoreline, Beaches, Sand Bars, Spits, Mud Flats & Salt Flats, Mangrove Submerged Roots
Co-Habitants

Northern Raccoon 
Green Turtle 
Tiger Shark 
Weedy Seadragon 
Vaquita
Behavior
Typically diurnal, except during the mating season, solitary hawksbill turtles comb the reefs and continental shelves searching for food during the daylight hours.
Hawksbill turtles were once thought to have remained in one local area for the duration of their lives, however, recent studies have proven that they migrate very long distances during their lifetimes. Hawksbill turtles are now known to be highly migratory individuals that undertake complex movements through geographically disparate habitats during their lifetimes. Once sexually mature, they undertake breeding migrations between a wide range of broadly separated localities, habitats, foraging grounds, and breeding areas at intervals of several years.
While most hawksbill turtles undertake long migrations, some portion of immature animals may settle into foraging habitats near their beaches of origin.
The mechanisms that aid hawksbill turtles in returning to their nesting beaches are still unknown. It has been thought that these turtles are guided inland by magnetic fields and the lunar phases and positioning of the moon.
CIRCADIAN RHYTHM
DiurnalMOVEMENT PATTERN
Full Migrant
Diet
Hawksbill turtles are omnivorous and feed primarily on sponges, extracting them from crevices on the reef by using their narrow, pointed beaks. Unusually amongst marine animals, to whom they are often unpalatable, sponges make up the majority of the hawksbill’s diet.
Hawksbill turtles show a large level of feeding selectivity in the way that they only eat certain species of sponges, some of which are toxic to other animals. They also prefer feeding grounds in shallow shoals abundant with brown algae.
Sea jellies and other coelenterates are also common prey items, as well as mollusks, fish, marine algae, crustaceans, and other sea plants and animals.
Hawksbill turtles are primarily spongivorous in the Caribbean, but more omnivorous in the Indo-Pacific. They consume relatively large amounts of algae in northern Australia, soft corals in the Great Barrier Reef region, and other combinations of forage depending on habitat.
PREFERENCE
SpecialistSTYLE
Forager
Reproduction
Hawksbill turtle mating occurs roughly every 2 to 3 years and occurs mainly in shallow waters.
Little information is available as to whether or not these turtles have life-long partners or are promiscuous, but research published in 2013 found revealed that these turtles are mainly monogamous and don’t tend to re-mate during the season.
In northeastern Australia, first breeding is estimated to occur at 31-36 years for females and 38 years for males.
Data on reproductive longevity in Hawksbills is limited, but becoming available with increasing numbers of intensively monitored, long-term projects on protected beaches. During the last decade, numerous individual Caribbean hawksbills have been recorded actively nesting over a period of 14-22 years. In the Indo-Pacific, Mortimer, Bresson, and Limpus have reported nesting over 17-20 years, comparable to other Chelonid turtles which range from 20 to 30 years.
The hawksbill turtle communicates by the use of ritual mating behaviors.
Copulation usually begins in shallow waters near the shore where males lie and wait for the females to return. At times, males have been seen following the females on shore. However, this behavior is rarely observed.
Upon reaching sexual maturity, a female hawksbill turtle will typically lay up to five clutches of around 100 to 140 eggs in a single breeding season and then wait a few years before nesting again. Saturation tagging of nesting females indicates that at most sites the average female hawksbill lays between three and five egg clutches during a single nesting season, with indications that newly recruited females lay fewer egg clutches, and possibly fewer clutches in the Arabian/Persian Gulf.
Hawksbill turtle nesting is much more dispersed than in other marine turtles, but individuals do tend to return to a particular beach season after season. Global population genetic studies have demonstrated the tendency of female hawksbill turtles to return to breed at their natal rookery, even though as juveniles they may have foraged at developmental habitats located hundreds or thousands of kilometers from the natal beach.
Hawksbill turtles nest on insular and mainland sandy beaches throughout the tropics and subtropics in at least 70 countries, although much nesting now only occurs at low densities. The entire nesting process takes roughly one to three hours and involves similar steps as most other species of sea turtles. The turtles come out of the sea and select a site in which to lay their eggs. They then clear the area and dig a pit in the sand. Next, they lay their eggs and proceed to fill in the pit with sand using their hind limbs. After the eggs have been laid and the site has been disguised, the female turtles return to the sea.
Sex determination of hawksbill turtles is thought to be temperature-dependent, as is the case with other sea turtles and reptiles, however not enough data is available to be sure this is true. Generally, eggs that are laid in cooler environments, less than 28.5°Celsius, tend to produce more males than females, and warm nests, greater than 30.3°Celsius, are known to hatch more females than males.
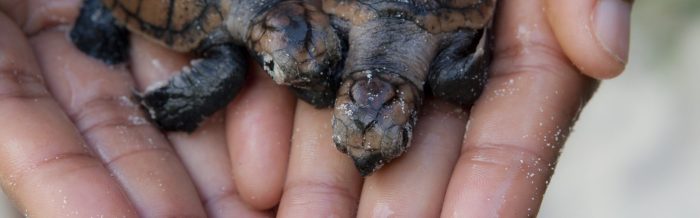
After about 60 days, newborn hawksbill turtles hatch out of their eggs and make a perilous dash for the water where they will mature. This journey to the water is the most dangerous time of a hawksbill turtle’s life. Although this scramble only lasts a few minutes, countless hatchlings are preyed on by flocks of gulls and large crabs. Probably less than one out of 1,000 eggs will survive and reach adulthood.
Having survived the dash to the sea, hawksbill turtle hatchlings are believed to spend their first few years in the open ocean before returning to more sheltered coastal waters. Recent studies indicate that the oceanic phase may be shorter for hawksbills, or even omitted in certain regions, as hatchlings swim less vigorously than those of other species. When hawksbill turtles are young, they are unable to dive into deep water, and therefore are forced to live in masses of floating sea plants, such as sargassum. Available data indicates that newly emerged hawksbill turtle hatchlings enter the sea and are carried by offshore currents into major gyre systems where they remain until reaching a carapace length of some 20 to 30 centimeters. At that point, they recruit into a neritic developmental foraging habitat that may comprise coral reefs or other hard-bottom habitats.
BREEDING INTERVAL
2-3 YearsBROOD
3-5PARENTAL INVESTMENT
NoneINCUBATION
60 DaysCLUTCH
100-140INDEPENDENCE
BirthSEXUAL MATURITY
31-38 Years
Ecology
Hawksbill turtles play a positive role in the ecosystem and help create habitats. They are important components of healthy coral reef ecosystems.
Like other species of sea turtles, hawksbills contribute to marine and coastal food webs and transport nutrients within the oceans. Oftentimes, they feed on sponges, causing succession to occur in the reef and freeing up space for settlement of other organisms. At sites where they are primarily spongivorous, hawksbills have been found to support healthy reefs by controlling sponges which would otherwise out-compete reef-building corals for space.
Adult turtles are consumed by humans, sharks, crocodiles, large fish, and octopi. Hawksbill turtle nests are also commonly robbed by terrestrial predators such as dogs, raccoons, rats, and humans.
There are no known adverse affects of the hawksbill turtle on humans, however, humans continue to harm these sea turtles and have become their major predator.
Although currently illegal throughout the world, humans have hunted the hawksbill turtle for years in order to sell and trade its scutes. Tortoiseshell, the beautiful scutes of the carapace and plastron of the hawksbill turtle, has been prized since ancient times. Surrounded by legend, tortoiseshell has been described as “one of the romantic articles of commerce, not only because of where it comes from, but because of the creatures from which it is obtained and the people engaged in the trade.” Jewelry and other tortoiseshell objects have been unearthed from pre-dynastic graves of the Nubian rulers of Egypt and excavated from the ruins of the Han Empire of China. Over 2,000 years ago, Julius Caesar considered tortoiseshell to be the chief spoil of his triumph. By the 9th Century, caravans of Arab traders carried rhinoceros horn, ivory, and tortoiseshell throughout the Indian Ocean. For the next 1,000 years, the tortoiseshell trade flourished. Around 1700, during the Edo Period, the bekko, (tortoiseshell,) artisans of Japan established themselves at Nagasaki. The tortoiseshell trade has been closely linked to European discovery, conquest, and commerce around the world. The Portuguese, Dutch, French, and English played major roles in the global trade. Exploitation occurred throughout the world’s tropical oceans, and especially in the East Indies, (modern day India, Indo China, Indonesia, Malaysia, and Philippines.) The East Indies were a major source of the shell of antiquity, as the rich waters have fittingly been called the world’s most productive seas for tortoiseshell. In the insular Pacific, international trade did not develop until the mid 19th Century, but once established, it took a tremendous toll on the region’s hawksbills. For the next 150 years, tortoiseshell was a prized commodity in the Pacific, first with the sandal-wooders and then with the whalers.
HANDICRAFTS, JEWELLERY, ETC.
Local, National, International
Predators

Human 
Dog 
Northern Raccoon
Conservation
The hawksbilll turtle’s current Critically Endangered assessment on the International Union for Conservation of Nature and Natural Resources (IUCN) Red List of Threatened Species clearly demonstrates the importance of protection in both terrestrial and marine habitats.
In 2001, the IUCN Red List Standards and Petitions Subcommittee upheld the Critically Endangered listing of the hawksbill, based on ongoing and long-term declines in excess of 80% within the time frame of three generations and ongoing exploitation. The Subcommittee review cited “convincing evidence of reductions in excess of 80% over the last three generations at many, if not most of the important breeding sites throughout the global range of the species”. Not surprisingly, those declines reflect the intensity of the tortoiseshell trade in the 20th Century.
Population
It is very difficult to classify exactly how endangered hawksbill turtles are because they are migratory and found all throughout the world . Also, since there is little knowledge of their early population levels, it is very hard to know how much the populations have declined.
In some places, they may be very scarce, and in others they may thrive.
Sea turtle population trends are best diagnosed using in-water abundance estimates coupled with estimates of demographic parameters, such as survival and recruitment possibilities. However, these data rarely exist for hawksbill turtle populations and so most assessments are based on evaluating nesting trends, which assumes a close correlation between population trends and nesting activity.
The size of a hawksbill turtle nesting population is defined as the average number of individual females nesting per year. In some cases, population numbers can be determined by saturation tagging of nesting females or by recording the total number of slaughtered nesters. More often, however, population estimates need to be derived from records of the total number of egg clutches laid during a season. Following the pattern of earlier status reviews, the present assessment calculates the annual number of nesting females by dividing the total number of egg clutches recorded, by three to five to produce a bracketed population estimate.
In many parts of the world, hawksbill turtle populations have continued to decline since the publication of the previous International Union for Conservation of Nature and Natural Resources (IUCN) Red List of Threatened Species assessment. Continuing losses in southeast Asia are of particular concern.
Although some relatively large populations still exist, especially in Australia, this is not inconsistent with long-term global or even regional population reduction over three generations, a point noted by the Subcommittee. Unlike previous reviews of the status of the hawksbill, the present assessment is quantitative and provides a numerical basis for the global listing of the species as “Critically Endangered”. The 2001 findings of the IUCN Red List Standards and Petitions Subcommittee are as valid today as they were six years ago.
Analysis of historic and recent published and unpublished accounts indicate extensive subpopulation declines in all major ocean basins over the last three hawksbill generations. Analyses of subpopulation changes at 25 Index Sites distributed globally show an 84 to 87% decline in number of mature females nesting annually over the last 3 hawksbill generations. Numerous populations, especially some of the larger ones, have continued to decline since the last assessment of the species.
Today, some protected populations are stable or increasing, but the overall decline of the species, when considered within the context of three generations, has been in excess of 80%.
THREATS
Hawksbills face multiple, severe threats including the tortoiseshell trade, egg collection, slaughter for meat, destruction of nesting habitat, destruction of foraging habitat, hybrization with other species, entanglement and ingestion of marine debris (including fishing gear,) and oil pollution.
The volume of the tortoiseshell trade has diminished, yet it remains active and substantial, and the Japanese bekko industry remains intact.
Analysis of historic and recent published and unpublished accounts indicate extensive subpopulation declines in all major ocean basins over the last three hawksbill turtle generations as a result of overexploitation of adult females and eggs at nesting beaches, degradation of nesting habitats, the taking of juveniles and adults in foraging areas, incidental mortality relating to marine fisheries, and degradation of marine habitats.
RESIDENTIAL & COMMERCIAL DEVELOPMENT
Tourism & Recreation AreasENERGY PRODUCTION & MINING
Oil & Gas DrillingBIOLOGICAL RESOURCE USE
Fishing & Harvesting Aquatic ResourcesHUMAN INTRUSIONS & DISTURBANCE
Recreational ActivitiesPOLLUTION
Industrial & Military EffluentsCLIMATE CHANGE & SEVERE WEATHER
Other Impacts
ACTIONS
International trade in the hawksbill turtle is banned amongst signatory nations by its listing on Appendix I of the Convention on International Trade in Endangered Species (CITES).
With protection, some populations have stabilized, and others are now increasing, most notably in the Caribbean. The increases documented in the Caribbean coincide with dramatic reductions in take on the foraging grounds of Cuba which have, in effect, spared tens of thousands of large hawksbills since the early 1990s. Such increases provide hope for the future, but unfortunately are still the exception rather than the rule. Similar results are needed elsewhere.
In order to succeed in keeping hawksbill turtles in existence, there must be cooperation among all nations that have hawksbill populations in their waters. Free exchange of information on the turtles is needed to ensure that all nations are aware of the best and most efficient ways of keeping hawksbill turtles in existence.
FaunaFacts
Video
SourceS
- Bjorndal, K. A. (1999, October 31). “Conservation of hawksbill sea turtles: Perceptions and realities”.
- Bjorndal, K. A., Bolten, A. B., & Chaloupka, M. Y. (2000). Green turtle somatic growth model: Evidence for density dependence. Ecological Applications, 10(1), 269-282.
- Bjorndal, K.A. & Jackson, J. B. C. (2003). Role of sea turtles in marine ecosystems-reconstructing the past. In: P. L. Lutz, J. A. Musick, & J. Wyneken (Eds.), Biology of Sea Turtles (Vol. 2) (pp. 259-273). Boca Raton, FL: CRC Press.
- Bouchard, S. S. & Bjorndal, K. A. (2000). Sea turtles as biological transporters of nutrients and energy from marine to terrestrial ecosystems. Ecology, 81, 2305-2313.
- Boulon Jr, R. (1994, August 17). Growth rates of wild juvenile hawksbill turtles, Eretmochelys imbricata, in St. Thomas, U.S. Virgin Islands. Copeia, 1994(3), 811-814.
- Bowen, B. W., Grant, W. S., Hillis-Starr, Z., Shaver, D. J., Bjorndal, K. A., Bolten, A. B., & Bass, A. L. (2007). Mixed-stock analysis reveals the migrations of juvenile hawksbill turtles (Eretmochelys imbricata) in the Caribbean Sea. Molecular Ecology, 16, 49-60.
- Bowen, B. W. & Karl, S. A. (1997). Population genetics, phylogeography, and molecular evolution. In: P. L. Lutz & J. A. Musick (Eds.), The Biology of Sea Turtles (pp. 29-50). Boca Raton, FL: CRC Press.
- Bugoni, L. (2014, January). “The biology of sea turtles: Volume III”. Marine Biology Reasearch, 10(1), 94-95.
- Carr, A., Carr, M. H., & Meylan, A. B. (1978). The ecology and migrations of sea turtles, 7: The West Caribbean green turtle colony. Bulletin of American Museum of Natural History, 162(1), 1-46.
- Chaloupka, M. Y. & Limpus, C. J. (2001). Trends in the abundance of sea turtles resident in southern Great Barrier Reef waters. Biological Conservation, 102, 235-249.
- Chaloupka, M. Y. & Musick, J. A. (1997). Age, growth, and population dynamics. In: P. L. Lutz & J. A. Musick (Eds.), The Biology of Sea Turtles (pp. 233-273). Boca Raton, FL: CRC Press.
- Diez, C. E., van Dam, R. P., & Archibold, G. (2002). In-water survey of hawksbill turtles at Kuna Yala, Panamá. Marine Turtle Newsletter, 96, 11-13.
- Dobbs, K. A., Miller, J. D., Limpus, C. J., & Landry Jr., A. M. (1999). Hawksbill turtle, Eretmochelys imbricata, nesting at Milman Island, northern Great Barrier Reef, Australia. Chelonian Conservation and Biology, 3(2), 344-361.Bugoni, L. (2014, January) “The biology of sea turtles: Volume III”. Marine Biology Reasearch, 10(1), Bjorndal, K. A. 1999. Conservation of Hawksbill Sea Turtles: Perceptions and Realities”
- Edelman, M. (2004). “Eretmochelys imbricata“. Animal Diversity Web.
- Ernst, C. H. & Barbour, R. W. (1992, November 17). Turtles of the World. Washington, District of Columbia: Smithsonian Institution Press.
- Ernst, C., Lovich, J., & Barbour, R. (1994). Turtles of the United States and Canada. Washington & London: Smithsonian Institution Press.
- Fitzsimmons, N. N., Tucker, A. D., & Limpus, C. J. (1995). Long-term breeding histories of male green turtles and fidelity to a breeding ground. Marine Turtle Newsletter, 68, 2-4.
- Wikimedia Foundation. (2018, September 29). Hawksbill sea turtle. Wikipedia.
- Hersh, K. (2016). “Chelonia mydas“, Animal Diversity Web (ADW).
- Hill, M. S. (1998). Spongivory on Caribbean reefs releases corals from competition with sponges. Oecologia, 117, 143-150.
- Hirth, H. F. (1997, August). Synopsis of the biological data on the green turtle: Chelonia mydas (Linnaeus 1758). Biological Report, 97(1), 1-120.
- Groombridge, B. & Luxmoore, R. (1989). The Green Turtle and Hawksbill (Reptilia: Cheloniidae): World Status, Exploitation and Trade. Lausanne, Switzerland: Secretariat of the Convention on International Trade in Endangered Species of Wild Fauna and Flora.
- León, Y. M. & Bjorndal, K. A. (2002). Selective feeding in the hawksbill turtle, an important predator in coral reef ecosystems. Marine Ecology-Progress Series, 245, 249-258.
- Limpus, C. J. (1992). The hawksbill turtle, Eretmochelys imbricata, in Queensland: population structure within a southern Great Barrier Reef ground. Wildlife Research, 19, 489-506.
- Limpus, C. J. & Miller, J. D. (2008, April). Australian Hawksbill Turtle Population Dynamics Project. Queensland, Australia: Environmental Protection Agency.
- Lutz, P. L. & Musick, J. A. (1997). The Biology of Sea Turtles. Boca Raton, FL: CRC Press.
- Märzhäuser, H. (2018, February 16). Too hot for turtle guys: Great Barrier Reef is dangerously warm for male green sea turtles. Deutsche Welle.
- McKinnon, J. M. (1975). Tomahawks, turtles and traders: A reconstruction in the circular causation of warfare in the New Georgian group. Oceania, 45(4), 290-307.
- Meylan, A. B. (1988). Spongivory in hawksbill turtles: A diet of glass. Science, 239, 393-395.
- Milliken, T. & Tokunaga, H. (1987, July). The Japanese Sea Turtle Trade 1970-1986: A Special Report prepared by TRAFFIC (Japan). Washington, DC: Center for Environmental Education.
- Mortimer, J. & Bresson, R. (1999). Temporal distribution and periodicity in hawksbill turtles (Eretmochelys imbricata) nesting at Cousin Island, Republic of Seychelles, 1971-1997. Chelonian Conservation and Biology, 3(2), 318-325.
- Mortimer, J. A., Collie, J., Jupiter, T., Chapman, R., Liljevik, A., & Betsy, B. (2002, April). Growth rates of immature hawksbills (Eretmochelys imbricata) at Aldabra Atoll, Seychelles (Western Indian Ocean). In: J. A. Seminoff (Ed.), Proceedings of the Twenty-Second Annual Symposium on Sea Turtle Biology and Conservation. Miami, FL: National Oceanic and Atmospheric Administration (NOAA) TTechnical Memorandum NMFS-SEFSC-503.
- Mortimer, J. A., Day, M., & Broderick, D. (2002, January). Sea turtle populations of the Chagos Archipelago, British Indian Ocean territory. In: A. Mosier, A. Foley, & B. Brost (Eds.), Proceedings of the Twentieth Annual Symposium on Sea Turtle Biology and Conservation. Miami, FL: National Oceanic and Atmospheric Administration (NOAA) TTechnical Memorandum NMFS-SEFSC-477.
- Mortimer, J. A. & Donnelly, M. (IUCN SSC Marine Turtle Specialist Group). (2008). “Eretmochelys imbricate”. The IUCN Red List of Threatened Species 2008: e.T8005A128.
- Mortimer, J. A., Meylan, P. A., & Donnelly, M. (2007). Whose turtles are they, anyway? Molecular Ecology, 16, 17-18.
- Mosier, A., Foley, A., & Brost, B. (2002). Proceedings of the Twentieth Annual Symposium on Sea Turtle Biology and Conservation. Miami, FL: National Oceanic and Atmospheric Administration (NOAA) TTechnical Memorandum NMFS-SEFSC-477.
- Musick, J. A. & Limpus, C. J. (1997). Habitat utilization and migration in juvenile sea turtles. In: P. L. Lutz & J. A. Musick (Eds.), The Biology of Sea Turtles (pp. 137-164). Boca Raton, FL: CRC Press.
- Parrish, A. & Goodman, K. (2006, June). Tagging and Nesting Research on Hawksbill Turtles (Eretmochelys imbricata) at Jumby Bay, Long Island, Antigua, West Indies: 2002 Annual Report. Wider Caribbean Sea Turtle Conservation Network (WIDECAST).
- Parsons, J. J. (1972). The hawksbill turtle and the tortoise shell trade. In: Pierre, G. (Ed.), Études de géographie tropicale offertes a Pierre Gourou (pp. 45-60). Mouton, Paris: Office de La Recherche Scientifique et Technique Outre-mer.
- Pilcher Dr, N. Hawksbill turtle (Eretmochelys imbricata). Wildscreen Arkive.
- Pope, C. H. (1939). Turtles of the United States & Canada. New York, NY: Alfred A Knopf.
- Richardson, J. I., Bell, R., & Richardson, T. H. (1999). Population ecology and demographic implications drawn from an 11-year study of nesting hawksbill turtles, Eretmochelys imbricata, at Jumby Bay, Long Island, Antigua, West Indies. Chelonian Conservation and Biology 3(2), 244-250.
- Ripple, J. (1996). Sea Turtles. Vancouver, Canada: Voyager Press.
- Seminoff, J. A. (2002, April). Proceedings of the Twenty-Second Annual Symposium on Sea Turtle Biology and Conservation. Miami, FL: National Oceanic and Atmospheric Administration (NOAA) TTechnical Memorandum NMFS-SEFSC-503.
- Turtle Trax. (2005, June 8). “The hawksbill turtle (Eretmochelys imbricata)”.
- van Dam, R.P. & Diez, C.E. (1997). Diving behavior of immature hawksbill turtles (Eretmochelys imbricata) in a Caribbean reef habitat. Coral Reefs, 16, 133-138.
- Witzell, W. N. (1983). Synopsis of biological data on the hawksbill turtle, Eretmochelys imbricata (Linnaeus, 1766). Rome, Italy: Food and Agricultural Organization of the United Nations (FAO) Fisheries Synopsis No. 137.
- World Wildlife Foundation (WWF). “Hawksbill turtle”.