PHYSIOLOGY | IMAGES | ETYMOLOGY | TAXONOMY | GEOGRAPHY | BEHAVIOR | DIET | REPRODUCTION | ECOLOGY | CONSERVATION | FAUNAFACTS | VIDEO | SOURCES

The southern grasshopper mouse is an omnivorous rodent from North America that mostly feeds on arthropods, such as scorpions and grasshoppers. It is notable for its wolf-like howls and immunity to scorpion venom, granting it the nickname, “Scorpion Mouse”. Due to its aggressive and cannibalistic nature, it is solitary and highly territorial.
Physiology
The southern grasshopper mouse is a small, robust species. Its body size averages between 9-13 centimeters (3.5-5.1 inches) for the head and body and 3-6 centimeters (1.2-2.4 inches) for the tail.
The short, broad tail is club-shaped and longer than half the length of its head and body at 31-57%. The tail length of southern grasshopper mice museum specimens made up an average of 33.9% of their body length. Their tail length measured an average of 4.58 ± 0.36 centimeters in females, and 4.50 ± 0.39 centimeters in males. Average tail length does not vary greatly between the sexes.
There is sexual dimorphism displayed in the physiology of the southern grasshopper mouse as females are heavier and longer than males.
Measurements of cranial volume of museum specimens of southern grasshopper mouse have shown that on average, females were heavier and longer than males, but their cranial volume was about 3% less than that of males. On average, female southern grasshopper mice are also able to deal with a larger body size without increasing their brain size, when compared to males.
The tail length of southern grasshopper mice museum specimens made up an average of 33.9% of their body length. Their tail length measured an average of 4.58 ± 0.36 centimeters in females, and 4.50 ± 0.39 centimeters in males. Average tail length does not vary greatly between the sexes.
There is still not enough evidence to explain the difference in body size and brain between males and females. It is possible that there are different demands on both sexes, which might contribute to this difference. These studies were also only done in the laboratory, and there might be sex-specific stresses in their natural habitat that influence brain growth. However, sexual dimorphism is quite common and has been observed in the central nervous system of other mammals.
Relative brain size in southern grasshopper mice tends to vary with food preferences. Brain size is usually smallest in folivores, larger in both granivores and insectivores, and largest in generalists.
Measurements of cranial volume of museum specimens of southern grasshopper mouse have shown that on average, females were heavier and longer than males, but their cranial volume was about 3% less than that of males. However, this was not a significant difference. This pattern of brain-body relationship can also be seen in northern grasshopper mice (Onychomys leucogaster).
On average, female southern grasshopper mice are able to deal with a larger body size without increasing their brain size, when compared to males. There is still not enough evidence to explain the difference in body size and brain between males and females. It is possible that there are different demands on both sexes, which might contribute to this difference. These studies were also only done in the laboratory, and there might be sex-specific stresses in their natural habitat that influence brain growth. However, sexual dimorphism is quite common and has been observed in the central nervous system of other mammals.
The southern grasshopper mouse is covered with fine, dense fur. The upper body is a grayish or pinkish-cinnamon color. The basal two-thirds of the tail are colored like the upper body, but the terminal tip is white like the underside of the body. The two colors are distinctly separated.
The southern grasshopper mouse has no specialized physiological adaptations to arid conditions. The southern grasshopper mouse drinks water in captivity, but probably obtains moisture from food under natural conditions.

BODY LENGTH
9-13 cm. / 3.5-5.1 in.TAIL LENGTH
3-6 cm. / 1.2-2.4 in.BODY MASS
22 g. / 0.8 oz.LIFESPAN
4.6 yr.GENERATION LENGTH
1-2 yr.LOCOMOTION
Plantigrade

Images
Taxonomy
The southern grasshopper mouse is a species of the order Rodentia, and is in the family Cricetidae. It is unique among other North American rodents in the family Cricetidae in that its diet consists mostly of arthropods.
The southern grasshopper mouse overlaps greatly in niche characteristics with the northern grasshopper mouse (Onychomys leucogaster), though it seems to prey more heavily on arthropods. In potential areas of overlap with the northern grasshopper mouse in southern Mono County, California, the southern grasshopper mouse may occupy areas of lower elevation. There are no other likely competitors.
Taxonomic accounts prepared by Hollister (1914) with a revision for Arizona by Van Cura and Hoffmeister (1966) described 10 distinct subspecies in the southern grasshopper mouse in 1975. O. torridus was described to be principally found in low, arid, scrub and semi-scrub vegetation of deserts in the Lower Sonoran Life-zone from western Nevada, southern California, and southwestern Utah, south to central Mexico, and east to western Texas. In areas of sympatry with the northern grasshopper mouse (Onychomys leucogaster), there was evidence that O. torridus occupied areas of lower elevation.
Seven subspecies are currently recognized by the Integrated Taxonomic Information System (ITIS): O. t. canus, O. t. clarus, O. t. perpallidus, O. t. ramona, O. t. surrufus, O. t. tularensis, and O. t. yakiensis.
KINGDOM
AnimaliaPHYLUM
ChordataCLASS
MammaliaORDER
RodentiaFAMILY
CricetidaeGENUS
OnychomysSPECIES
torridusSUBSPECIES (7)
O. t. canus, O. t. clarus, O. t. perpallidus, O. t. ramona, O. t. surrufus O. t. tularensis, O. t. yakiensis
Etymology
The southern grasshopper mouse is also known as the scorpion mouse. Both names are derived from its carnivorous diet consisting mostly of arthropods, especially scorpions and orthopteran insects, such as grasshoppers.
Southern grasshopper mice are most notable for killing and eating Arizona bark scorpions (Centruroides sculpturatus), a species with a highly venomous sting.
ALTERNATE
Scorpion MouseGROUP
Nest
Region
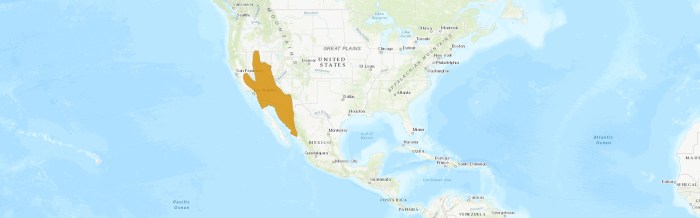
The southern grasshopper mouse is found mainly in the Western and Southwestern United States and in Northern Mexico of North America.
The species occurs from western Nevada, southern California, and southern Arizona in the United States, south to northern Baja California, Sonora, and northern Sinaloa in Mexico. They commonly inhabit the Mojave Desert and southern Central Valley of California.
EXTANT
Mexico, United States
Habitat
The southern grasshopper mouse inhabits temperate shrubland and desert habitats. It is found in shortgrass prairies; desert scrub; rocky soils; creosote, hot, arid valleys; and semi-desert areas.
Alkali desert scrub and desert scrub habitats are preferred, with somewhat lower densities expected in other desert habitats, including succulent shrub, wash, and riparian areas. Southern grasshopper mice also occur in coastal scrub, mixed chaparral, sagebrush, low sage, and bitterbrush habitats. They frequent desert areas, especially scrub habitats with friable soils for digging.
Southern grasshopper mice are uncommon in valley foothill and montane riparian, and in a variety of other habitats.
The most prefer xeric areas are at low elevations. Low to moderate shrub cover is preferred.
SHRUBLAND
TemperateDESERT
Temperate
Co-Habitants
Behavior
Southern grasshopper mice are largely nocturnal and active year round. They are terrestrial and good climbers.
Southern grasshopper mice often commit acts of cannibalism, killing and eating other members of their species if they are threatened or in need of food. As such, they are solitary animals. They may, however, live in male-female pairs year-round, although this seems to shorten the lifespan, as one of the two partners inevitably kills the other.
Perhaps the most unusual thing about the southern grasshopper mouse is its trademark howl. The mouse can produce a loud, piercing, pure tone, which lasts between 0.7 and 1.2 seconds and is audible to the human ear up to 100 meters away. Standing on their hind legs with their noses pointed upwards, they give this call when faced with an adversary, including other grasshopper mice, or prior to making a kill. This shrill warning, often repeated, is compared to a miniature wolf call, due to its smoothness and prolongation and to the animal’s wolf-like posture.
As with all predators, southern grasshopper mice require a large territory of two to three hectares in order to find the amount of food they require to survive. The territory size may equal the home range and may occur in male-female pairs.
A male and female along with their offspring occupy a larger home range for burrowing compared to males and females alone. On average, males have a home range of 7.8 acres, while females occupy an area of 5.9 acres.
The home range of southern grasshopper mice males in the Chihuahuan Desert during the non-breeding season was observed to be smaller compared to the home range of males during the breeding season. In contrast, there was not a significant difference between the home ranges of females during the breeding and non-breeding seasons. The average home range of adult males was slightly greater, about two to three times bigger, compared to adult females during the reproductive season. Although this is a big difference, these results were also not very significant. The larger home range of males during the reproductive season might be a strategy they use to maximize the number of encounters they could have with potential female mates when trying to reproduce.
The home ranges between southern grasshopper mice are not too far from each other, with an average maximum distance of 118.4± 8.0 meters, but are widely separated from neighbors.
Adult males tend to be highly territorial and guard their large territories fiercely against all intruders. Their efficient spacing is facilitated by high-pitched calls and vocalizations made at night to serve as territorial advertisement.
Southern grasshopper mice tend to maintain a specific place of residency for a longer period of time compared to other mammals.
The southern grasshopper mouse nests in small burrows dug into the ground. Often, these burrows have been deserted by other rodents or were taken by the grasshopper mouse through force. They may also be excavated.
The southern grasshopper mouse is not migratory and does not have seasonal movements.
CIRCADIAN RHYTHM
NocturnalMOVEMENT PATTERN
Non-Migrant
Diet
Southern grasshopper mice are omnivorous, but have a mainly carnivorous diet, feeding almost entirely on arthropods. Both vertebrates and seeds are minor components of the diet.
10-25% of the southern grasshopper mouse’s diet consists of seeds, plants, and vegetables. Less than 5% is seeds.
The remaining 75-90% includes mainly scorpions, but also grasshoppers, beetles, and small vertebrates, including salamanders, lizards, frogs, small mammals, and other rodents, such as Peromyscus, Perognathus, Microtus, the Los Angeles little pocket mouse (Perognathus longimembris), and the southern marsh harvest mouse (Reithrodontomys megalotis). In 1929, laboratory studies revealed the diet was composed of 56% grasshoppers, crickets, caterpillars, and moths, and 21% ground and darkling beetles.
The southern grasshopper mouse is an extremely aggressive predator and hunts its prey like most sophisticated predators. After stalking its potential kill, the grasshopper mouse will seize the animal with a rush, killing with a bite to the head. While overpowering its prey, the mouse closes its eyes and lays its ears back.
In a study done by Richard McCarty and Charles H. Southwick, southern grasshopper mice were deprived of food and their predatory behavior against laboratory mice and crickets was observed. After 48 hours of food deprivation, the mice spent more time eating crickets than the laboratory food they were provided. The crickets were both dead and alive, but the mice spent more time eating the unharmed living crickets, which might suggest that prey movement might be a catalyst and cue for predatory attack. The difference between the time that females and males spent eating living crickets was not significant, indicating that predatory attack is not necessarily more dominant in just one sex. When the mice came in contact with laboratory mice of both sexes, they also showed very aggressive behaviors, such as biting the back and tail of their prey. After two days of being around the laboratory mice, six of the 43 male grasshopper mice and 22 of the 45 females grasshopper mice killed and ate certain parts of the laboratory mice. The difference between this aggressive behavior between the male and female grasshopper mice was significant. The study concluded that food deprivation and the type of prey affected the intensity and aggressiveness of the predatory attacks observed by southern grasshopper mice.
The southern grasshopper mouse is notable for its resistance to venom as it routinely kills and eats Arizona bark scorpions (Centruroides sculpturatus), a species with a highly venomous sting. In the arid regions inhabited by the southern grasshopper mouse, the Arizona bark scorpion is plentiful and avoided by most predators because of the very painful sting it can inflict. The grasshopper mouse can feed on this scorpion with impunity even when the scorpion stings it repeatedly in the face.
Researchers have found that a neural mechanism is involved that blocks the sending of pain messages to the brain. This mouse can normally feel pain from other sources, but can be temporarily insensitive to these stimuli after a dose of venom from a bark scorpion. This pain modulation is activated soon after the scorpion sting. For a moment, the mouse feels pain, but then the scorpion venom binds to the transmembrane channel otNav1.8. Specifically, the venom binds to a glutamic acid residue located within the channel protein. This prevents neuron action potentials from firing, and acts as a type of temporary anesthetic and pain reliever for the mouse. When this glutamic acid was replaced with hydrophilic glutamine residue, the venom did not bind with the protein, and the pain modulation ability was lost.
PREFERENCE
GeneralistSTYLE
Ambush
Prey
Reproduction
The southern grasshopper mouse is usually known to be a monogamous species. Both males and females care for the young.
Southern grasshopper mice are capable of breeding year-round, but peak reproductive activity occurs during the late spring and summer from May to July. Breeding may start in January under ideal conditions.
Female southern grasshopper mice become receptive at 6 weeks of age and give birth to their first litter when four or five months old. Gestation lasts 26-35 days, with an average of 29 days. Litter size ranges from one to six young with an average litter size of 2.6-4.
Young females apparently have significantly greater reproductive potential than old females. They are able to produce as many as six litters in a year in the wild, but their reproductive life is short, few breeding successfully in their second year. Females tend to be sexually active only during one breeding season, which is due to their rapid reproductive aging following their first year. Female southern grasshopper mice born as early as April may produce two or three litters before the year’s end, but females born in late summer may have as many as six litters during the following breeding season.
Young southern grasshopper mice are born naked with pink translucent skin and weigh approximately 2.6 grams, or 0.09 ounces. One day later, the pigment develops on the dorsal surface and by day 10, hairs of 3 millimeters or 0.12 inches long are present on the back.
The young mice immediately begin nursing from their mother and open their eyes on day 15, at two weeks of age. They are weaned at three weeks of age and begin consuming solid food by day 19. Weaning in the laboratory may occur in 20 days.
BREEDING SEASON
Spring-SummerBREEDING INTERVAL
2-6 MonthsPARENTAL INVESTMENT
Maternal, PaternalGESTATION
26-35 DaysLITTER
1-6WEANING
3 WeeksSEXUAL MATURITY
6 Weeks
Ecology
Southern grasshopper mice, despite their small size, do not have too many predators. Owls and other nocturnal birds of prey usually feed on these rodents. Even snakes, centipedes, and scorpions avoid consuming these rodents as they’re known to be prone to venom and difficult to catch.
However, human activities do have an impact on the habitat of the mice. Other potential threats include urbanization and industrialization by humans.
As with many rodents, southern grasshopper mice are extremely fertile. They are difficult to wipe out, even by humans.
Largely due to their ferocity and appetite, southern grasshopper mice are considered beneficial by farmers and others because they eat potentially harmful insects. They help farmers by eating a large number of insects and rodents that would otherwise destroy crops.
Although southern grasshopper mice are mainly carnivorous, they do eat the same crops they normally help defend if their usual prey, insects and small vertebrates, become scarce.
Predators
Conservation
The southern grasshopper mouse is listed as Least Concern on the International Union for Conservation of Nature and Natural Resources (IUCN) Red List of Threatened Species in view of its wide distribution, presumed large population, and because it is unlikely to be declining at nearly the rate required to qualify for listing in a threatened category.
The southern grasshopper mouse has a currently stable population trend and the population is not severely fragmented.
Population
The southern grasshopper mouse is fairly common throughout its range, but is usually found in areas with low population densities.
Several studies have demonstrated that the reproductive attributes of this species allows them to be naturally maintained at lower population densities. Females tend to be sexually active only during one breeding season, which is due to their rapid reproductive aging following their first year. Moreover, males’ unique pauses in their testicular activity during the breeding season might also contribute to the lower population densities in which they live. Males store sperm at 40 days of age.
Home ranges of grasshopper mice in New Mexico were 3.2 hectares (7.8 acres) for males, and 2.4 hectares (5.9 acres) for females (Blair 1943). In southeast Arizona, the average home range of adult mice was 11.45 hectares (28 acres) (Chew and Chew 1970). In Nevada desert scrub, density averaged 1.83 mice per hectare (0.74 mice/acre).
FRAGMENTATION
Not Fragmented
THREATS
The species has no known major threats. There are no currently known conservation issues associated with southern grasshopper mice, which is mainly due to its wide distribution and tolerance for disturbances caused by arthropods. However, some disturbances have been observed in their low density populations.
ACTIONS
There are no known conservation measures specific to the southern grasshopper mouse.
However, there are several protected areas within its range. Conservation sites have been identified over the species’ entire range.
Many more population studies will need to be performed in order to gain further insight into the conservation status of this species.
FaunaFacts
Video
SourceS
- Brylski, P. Southern grasshopper mouse. California Wildlife Habitat Relationships System: California Department of Fish and Wildlife.
- Chew, R. M. & Chew, A. E. (1970). Energy relationships of the mammals of a desert shrub (Larrea tridentata) community. Ecological Mongorraphs, 40(1), 1-21.
- Frank, D. H. & Heske, E. J. (1992). Seasonal changes in space use patterns in the southern grasshopper mouse, Onychomys torridus torridus. Journal of Mammalogy, 73(2), 292-298.
- Gennaro, A. L. (1968). Northern limits of four desert rodents of the genera Peromyscus, Perognathus, Dipodomys and
- Onychomys in the Rio Grande Valley. The American Midland Naturalist Journal, 80, 477-493.
- Grzimek, B. (1990). Grzimek’s Encyclopedia of Mammals (Vol. 5). New York, NY: McGraw-Hill Publishing Company.
- Guevara-Carrizales, A. A., Escobar-Flores, J., & Martínez-Gallardo, R. (2012). Marginal record of the southern grasshopper mouse Onychomys torridus in Baja California, México. Western North American Naturalist, 72(3), 416-417.
- Hall, E. R. & Kelson, K. R. (1959). The Mammals of North America. New York, NY: Ronald Press Co.
- Horner, B. E. & Taylor, J. M. (1968). Growth and reproductive behavior in the southern grasshopper mouse. Journal of Mammalogy, 49, 644-660.
- Horner, B. E., Taylor, J. M., & Padykula, H. A. (1964). Food habits and gastric morphology of the grasshopper mouse. Journal of Mammalogy, 45, 513-535.
- ITIS. (2020, October 1). Onychomyus torridus (Coues, 1874). Integrated Taxonomic Information System (ITIS).
- Kester, D. (1999). Onychomys torridus. Animal Diversity Web.
- Lacher, T., Timm, R., & Álvarez-Castañeda, S. T. (2016). Onychomys torridus (errata version published in 2017). The IUCN Red List of Threatened Species 2016: e.T15339A115127523. https://dx.doi.org/10.2305/IUCN.UK.2016-3.RLTS.T15339A22354700.en.
- Macdonald, D. (1995). The Encyclopedia of Mammals. New York, NY: Facts on File, Inc.
- Mann, M. D. & Towe, A. L. (2003). Brain-body size relations in grasshopper mice. Brain, Behavior and Evolution, 62(1), 13-18.
- McCarty, R. (1975). Onychomys torridus. Mammalian Species, 59, 1–5. doi:10.2307/3503863. JSTOR 3503863.
- McCarty, R. & Southwick, C. H. (1981). Food deprivation: Effects on the predatory behavior of southern grasshopper mice (Onychomys torridus). Aggressive Behavior, 7(2), 123-130.
- Nowak, R. M. (1999). Walker’s Mammals of the World (6 ed., Vol. 2). Baltimore: MD: John Hopkins University Press.
- Reardon, S. (2013, November 1). Zoologger: Mouse eats scorpions and howls at the moon. NewScientist.
- Thompson, B. (2018). The grasshopper mouse and bark scorpion: Evolutionary biology meets pain modulation and selective receptor inactivation. Journal of Undergraduate Neuroscience Education, 16(2), R51-R53.
- Wade, L. (2013, October 24). Mouse impervious to scorpion’s sting. Science.
- The Wikimedia Foundation. (2020, May 9). Southern grasshopper mouse. Wikipedia.
- Wilson, D. E. & Reeder, D. M. (2005). Mammal Species of the World: A Taxonomic and Geographic Reference (3 ed.). Baltimore, MD: Johns Hopkins University Press.