PHYSIOLOGY | IMAGES | ETYMOLOGY | TAXONOMY | GEOGRAPHY | BEHAVIOR | DIET | REPRODUCTION | ECOLOGY | CONSERVATION | FAUNAFACTS | VIDEO | SOURCES

The weedy seadragon is a common seadragon species found along the coast of Southern Australia. This colorful species has several leaf-like appendages that allow it to camouflage with floating seaweed as it sucks up its small shrimp, crustacean, and zooplankton prey through its pipe-like terminal mouth.
Physiology
Weedy sea dragons are sexually dimorphic, as males have narrower bodies and are darker than females.
A weedy sea dragon reaches 45 centimeters, or 18 inches, in length and has a narrow body. The body of the weedy seadragon is surrounded by protective dermal plates, which inhibits its mobility. Weedy seadragons lack a caudal fin, but have a long dorsal fin along the back and small pectoral fins on either side of the neck. The weedy seadragon also has two spines above its eye, one in front of the eye, for protection.
Weedy seadragons have no teeth, but instead have a long, tubular snout. This pipe-like, terminal mouth has an intricate system of bones pulled by muscles.
Adult weedy seadragons are often a reddish color with yellow and purple markings. The bodies of these fish are usually red with yellow spots. The beautiful colors of this seadragon are highlighted by the seven iridescent blue-violet bands along the upper body near the head.
Though they are similar in appearance to seahorses, which use a prehensile tail to clasp and anchor themselves to seaweed, weedy seadragons do not have prehensile, gripping tails. Instead, weedy seadragons appear to use their tails for steering.

SEXUAL DIMORPHISM
Narrower, Darker MalesBODY LENGTH
45 cm. / 18 in.LIFESPAN
10 yr.

Images
Taxonomy
The weedy seadragon is a marine fish related to the seahorse. It’s in the subfamily Syngnathinae, which contains all pipefish.
The weedy seadragon has no recognized subspecies.
The weedy seadragon is most closely related to the other member of its genus, the ruby seadragon (Phyllopteryx dewysea) and also the leafy seadragon (Phycodurus eques). The ribboned pipefish (Haliichthys taeniophorus) is not closely related to the weedy seadragon and does not form a true monophyletic clade with weedy and leafy seadragons.
The weedy seadragon was previously the only member of its genus until the description of the ruby seadragon in 2015.
Because of its similar appearance, the weedy seadragon is often mistaken for its close relative, the leafy seadragon (Phycodurus eques). The leafy seadragon is found in the same geographic range as the weedy seadragon, but it differs in appearance as it has many more leaf-like appendages. Weedy seadragons are also much more common than leafy seadragons.
KINGDOM
AnimaliaPHYLUM
ChordataCLASS
ActinopterygiiORDER
SyngnathiformesFAMILY
SyngnathidaeGENUS
PhyllopteryxSPECIES
TaeniolatusSUBSPECIES
None
Etymology
The weedy seadragon is named for its varying number of small, leaf-like appendages, either paired or single, along its body. These purple appendages have a black border and provide the fish camouflage and protection in its habitat because they resemble floating kelp fronds and seaweed.
ALTERNATE
Common Seadragon, Lucas’ Seadragon
Region
The weedy seadragon is endemic to the Australian waters of the Eastern Indian Ocean and the South Western Pacific Ocean.
It can be found along much of the southern Australian coastline, from near Newcastle and Port Stephens, New South Wales (approximately 32° 56’S) southwards to Actaeon Island, Tasmania (43°32’S) and westwards through Victoria and South Australia to Geraldton, Western Australia (28°46’S).
Individuals of this species have been sighted off the eastern coast of Australia in New South Wales, as far north as Port Stephens; along the southern coast; and up around the western coast of Australia as far north as Geraldton, West Australia. They have also been spotted near Rottnest Island, Western Australia.
EXTANT
Australia
Habitat
The weedy seadragon is a coastal marine animal that inhabits rocky reefs, seaweed beds, sea grass meadows, kelp gardens, and sandy areas. They occur over rocky reefs with algae supporting strands of kelp or other macroalgae or over adjacent stretches of seagrass and sand where they feed on mysids and small crustaceans.
The weedy seadragons’ depth distribution ranges from shallow bays down to reefs at depths of up to 50 meters (164 feet). Most weedy seadragons are found below the low tide line.
Although weedy seadragons have a broad range of habitat, they have very specific requirements. The water must be between 12 and 23 degrees Celsius (53-73 degrees Fahrenheit), and 8-50 meters deep (26-164 feet), although they most often are found between 8 and 12 meters deep (26-39 feet).
Seagrass and algal affiliations have been recorded with the species Halophila ovalis, Ecklonia radiata, Macrocystis pyrifera, M. angustifolia, Posidinia spp., Amphibolis spp., and Sargassum spp. Weedy seadragons also live in association with sponges.
Individuals have been shown to have broad, overlapping home ranges that can vary in length from 50–150 meters, and be up to 50 meters wide.
MARINE NERITIC
Pelagic, Macroalgal/Kelp, Submerged Seagrass
Co-Habitants
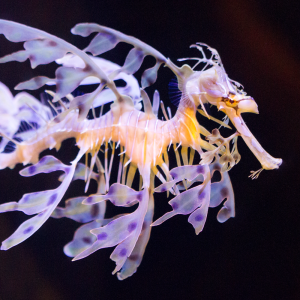
Leafy Seadragon 
Long-Spined Sea Urchin
Behavior
Most of the time, weedy seadragons are solitary, and are observed on their own or in pairs. However, larger grouping has been observed. In the Sydney area and in southern New South Wales, aggregations of between 20 and 40 seadragons have been observed, respectively.
Weedy seadragons are not sessile or immobile, but they are not very good swimmers, either. This is because their bodies are surrounded by protective dermal plates, which inhibit their mobility. Also, they lack a caudal fin, and therefore must rely on their ventral and dorsal fins for swimming. Weedy seadragons do, however, have a long dorsal fin along the back and small pectoral fins on either side of the neck. These fins help provide balance. Because they are poor swimmers, each year a number of individuals are found washed ashore on the beaches of southern Australia.
Because weedy seadragons are not good swimmers, they are slow-moving and drift in the water with their life-like appendages resembling the swaying seaweed of their habitat. As such, the weedy seadragon’s primary defense mechanism against predation is its camouflage abilities.
Most weedy seadragons live for up to six years, but increased longevity as been documented at some latitudes. The von Bertalanffy growth curve parameters for the weedy seadragon vary based on latitude, and are as follows: Sydney: L00=36.28 cm, L0=3.33 cm, and K=1.52; Hobart: L00=34.07 cm, L 0=3.2 cm, and K=0.91. Also, depending on where they are found latitudinally, the species can exhibit variable growth and survival rates. Slower growth and high survival, as exhibited at higher latitudes, lead to increased longevity, and in the more southerly portion of its range this species is suspected to have a longer lifespan, likely exceeding 10 years.
CIRCADIAN RHYTHM
DiurnalMOVEMENT PATTERN
Non-Migrant
Diet
The weedy seadragon is a carnivore and feeds on mysid shrimps, carid shrimps, prawns of the genus Lucifer, sea lice, larval fish, zooplankton, and other small crustaceans.
Weedy seadragons have no teeth, but instead feed by way of suction. Their pipe-like terminal mouth has an intricate system of bones pulled by muscles to create a strong suction force that is directed at food.
PREFERENCE
GeneralistSTYLE
Forager
Reproduction
Although the weedy seadragon’s reproductive strategies are well documented, researchers have yet to understand what biological or environmental factors trigger them to reproduce.
Weedy seadragon mating and pregnancies in captivity are rare. The Aquarium of the Pacific in Long Beach, California and the Tennessee Aquarium in Chattanooga, Tennessee in the United States of America, and the Melbourne Aquarium in Melbourne, Australia are among the few facilities in the world to have successfully bred weedy seadragons in captivity, though others occasionally report egg laying. In March 2012, the Georgia Aquarium in Atlanta, Georgia of the United States of America announced a successful breeding event of weedy seadragons. As of July 2012, the Monterey Bay Aquarium has also successfully bred and hatched baby weedy seadragons on exhibit.
The breeding season for weedy seadragons is generally July to January, but reproduction occurs later, October to March, in Tasmanian waters. Some individuals mature in one year, but most usually breed in their second year when fully grown. Weedy seadragons usually take about 28 months to reach sexual maturity.
Like their relatives the seahorses, male weedy seadragons carry and brood the developing eggs externally below their tail. Seadragons, seahorses, and pipefish are the only species in which the male carries the eggs. Brooding males have been observed in New South Wales from mid-winter to mid-summer, but never from February to June, despite sightings of over 350 seadragons during this period. A more recent study has confirmed this and also indicated that some males exhibit multiple pregnancies in a season. Males brood two batches of eggs per breeding season, and females are also able to produce multiple broods per season.
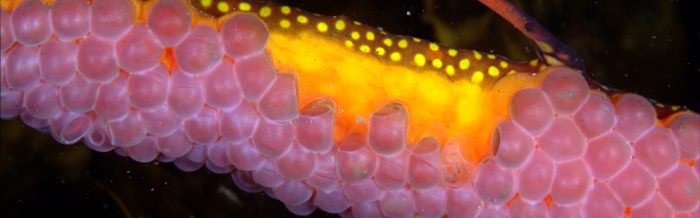
When a male is ready to receive the eggs, which he indicates by wrinkling the lower half of his tail, the female deposits about 120-250 ruby-colored eggs onto his brood patch. These eggs look like small, red grapes. The skin of the brood patch is made of blood-rich tissue and will form a cup around each egg during deposition. Each cup holds and nourishes one egg. After the eggs are deposited, they are fertilized by the male. After an incubation time of about eight weeks, the eggs hatch over a period of a couple days.
After the female weedy seadragon deposits the eggs into the male’s brood patch and the male carries the eggs until they hatch, the young receive no parental care because they are released into the external environment.
The survival rate for young weedy seadragons is low in the wild, but is about 60% in captivity. Although weedy seadragons lay 120-250 eggs, only 60-120 offspring will survive, while the others will fall prey to sea anemones. After hatching, young weedy seadragons spend two or three days in the yolk sac of the egg, where they continue to be nourished. After the young leave the yolk sac, they are independent and begin feeding on copepods and rotifers shortly after.
BREEDING SEASON
July-MarchBROOD
2PARENTAL INVESTMENT
NoneINCUBATION
2 MonthsCLUTCH
150-250INDEPENDENCE
BirthSEXUAL MATURITY
1-2 Years
Ecology
The peaceful weedy seadragon does not in any way negatively affect the human species. In fact, many people go scuba diving off the coast of southern Australia specifically to see weedy seadragons, which, therefore, promotes tourism.
The weedy seadragon is the marine emblem of the Australian State of Victoria. Victoria established a program to select a marine icon species for Victoria. This was an exciting opportunity to engage Victorians in learning about local marine life and to provide an opportunity to participate in the selection of an appropriate emblem for the rich and diverse marine environment. The weedy seadragon was selected as Victoria’s official marine faunal emblem by public nomination and was proclaimed by the Governor in Council on October 31, 2002.
The weedy seadragon is exploited for the aquarium trade at low levels that are not likely of conservation concern. The volume of wild-caught individuals is small and therefore not currently a major threat.
Unlike other syngnathids, the weedy seadragon is not presently a victim of bycatch or a target of trade in traditional Chinese medicine. These activities are currently a threat to many related seahorse and pipefish populations, as members of these species have often been used in Asia as aphrodisiacs and other medicines. Oriental herbalists can sell their dried and powdered bodies for up to $200 per gram.
PETS/DISPLAY ANIMALS, HORTICULTURE
National, International
Conservation
The weedy seadragon is threatened by habitat degradation and loss due to pollution and sedimentation, especially in urban areas. A large portion of the species’ range does however occur in some less populated areas that are not at risk. Therefore this species is listed as Least Concern on the International Union for Conservation of Nature and Natural Resources (IUCN) Red List of Threatened Species.
In 1996, the weedy seadragon was classified as Data Deficient, and in 2006, it was listed as Near Threatened.
Population
To date there has been no range-wide population estimate made for weedy seadragons, but several local studies have occurred.
One study conducted at five sites near Sydney and Hobart from 2001-2007 and 2003-2004, respectively, found densities of 10 to 70 animals per hectare, with significant declines at three of the five sites. These declines however were not directly attributable to anthropogenic causes, but it is likely given that pollution and invasive species are of concern in the area.
Further research is needed to quantify human impacts on the species and to tease them apart from natural cycles and disease outbreaks.
THREATS
The weedy seadragon is primarily threatened by habitat degradation and loss due to pollution and sedimentation, especially in urban areas. The loss of suitable seagrass beds, coupled with natural history traits that make them poor dispersers, put the future of seadragon populations at risk.
Weedy seadragons are killed by pollution and fertilizer run-off in their shallow, coastal habitats. Both algae-covered reefs and seagrass meadows have been adversely affected by human activities and loss in quality and quantity of habitat has been documented within its range.
The degradation of habitat is worst near major urban centers, such as Sydney, Adelaide, Melbourne, Perth, and Hobart, where discharge of storm water and treated sewage leads to eutrophication and increased sedimentation. Losses of seagrass have been particularly severe along the metropolitan coasts and are well documented. Reefs have been more strongly affected by increased rates of sedimentation resulting from human activities such as sand dredging, land reclamation and coastal urban development.
It is suspected that habitat degradation is playing a role in declines in weedy seadragons being observed along urban coasts, but further research is needed in order to confirm this. Further population and habitat monitoring are needed to decisively link habitat degradation to population declines.
Invasive species including urchins that degrade kelp habitat are an issue for weedy seadragons and may be contributing to declines. In Tasmania, the macroalgae-covered reefs on which seadragons occur face another threat. Over the last 20 years, the sea urchin Centrostephanus rodgersii, has become more common and increased its range around the state. The increase in sea urchins is considered to have resulted from harvesting of rock lobster, a known predator of urchins, and climate change. Grazing by urchins reduces the abundance of kelp and other macroalgae through the formation of urchin barrens.
Significant losses of giant kelp (Macrocystis pyrifera) have been documented over the past 20 years, associated with increased water temperatures. The reduction in macroalgae could adversely affect weedy seadragons although this has not been demonstrated.
RESIDENTIAL & COMMERCIAL DEVELOPMENT
Housing & Urban Areas, Commercial & Industrial AreasPOLLUTION
Domestic & Urban Waste Water, Industrial & Military Effluents, Agricultural & Forestry EffluentsCLIMATE CHANGE & SEVERE WEATHER
Habitat Shifting & Alteration
ACTIONS
Along with all syngnathids, the weedy seadragon is protected from exploitation by the Australian Environment Protection and Biodiversity Conservation Act of 1999.
There are no species-specific conservation measures in place for the species, but they are legally protected in both New South Wales and Tasmania and are subject to strict export controls. It is illegal to take or export weedy seadragons in most of the states within which they occur.
This species also occurs in several protected areas, such as Botany Bay, and is not mentioned in any international legislation or trade regulations.
A database of weedy seadragon sightings, as well as other seadragon species, known as Dragon Search has been established with support from the Marine and Coastal Community Network (MCCN), Threatened Species Network (TSN), and the Australian Marine Conservation Society (AMCS), which encourages divers to report sightings.
Monitoring of populations may provide indications of local water quality. Weedy seadragons could also become an important flagship species for the often-overlooked richness of the unique flora and fauna of Australia’s south coast.
To date, there has been no range-wide population estimates made for the weedy seadragon, but several local studies have occurred. Further research and monitoring are needed in order to determine population size and trends in abundance for this species.
One study conducted at five sites near Sydney and Hobart from 2001-2007 and 2003-2004, respectively, found densities of 10 to 70 animals per hectare, with significant declines at three of the five sites. These declines however were not directly attributable to anthropogenic causes, but it is likely given that pollution and invasive species are of concern in the area. Further research is needed to quantify human impacts on the species and to tease them apart from natural cycles and disease outbreaks.
In South Australia, a possible contraction in area of occupancy has been mentioned. Historical records exist from benthic surveys in Gulf St. Vincent in the 1965–1971 period, where reportedly “numerous weedies were observed” in northern Gulf St. Vincent, adjacent to the city of Adelaide. No sightings in this area have been reported to Dragon Search during the late 1990s and early 2000s.
Admittedly the lack of recent sightings might be due to the lack of popular diving spots in the northern gulf, and the lack of systematic surveys in recent years. However, the occurrence of seadragons might have been affected by significant habitat degradation and loss recorded since the 1960s in this part of the gulf. In particular, large areas of seagrass have been lost in waters deeper than 10 meters south of a line between Ardrossan and Port Prime.
FaunaFacts
Video
SourceS
- The Associated Press. (2008, June 12). Endangered sea dragon at Ga. aquarium pregnant. Fox News.
- Baker, J. L. (2009). Dragon Search: Public Report – Summary of National Sighting Data, 1990 to 2005. Report for Dragon Search Community – Based Monitoring Program. Adelaide, South Australia: Australian Marine Conservation Society: Reef Watch.
- Binohlan, C. B. (2018). Phyllopteryx taeniolatus (Lacepède, 1804): Common seadragon. FishBase.
- Bray, D. J. (2011). Common seadragon, Phyllopteryx taeniolatus (Lacépède 1804). Fishes of Australia.
- Dawson, C.E.( 1985). Indo-Pacific Pipefishes: Red Sea to the Americas. MS: USA: The Gulf Coast Research Laboratory Ocean Springs.
- Dragon Search. (2000). A community environmental monitoring project based in South Australia. Conservation South Australia.
- Edyvane, K. (2003). Conservation, Monitoring and Recovery of Threatened Giant Kelp (Macrocystis pyrifera) Beds in Tasmania: Final Report to Environment Australia. Hoabrt, Australia: Department of Primary Industries, Water & Environment.
- Forsgren, K. L. & Lowe, C. G. (2006). The life history of weedy seadragons, Phyllopteryx taeniolatus (Teleostei : Syngnathidae). Marine and Freshwater Research, 57(3), 313-322.
- Frostic, A. (2000). Phyllopteryx taeniolatus. Animal Diversity Web.
Glover, T. D., Southcott, C. J., & Scott, R. V. (1974). The Marine and Freshwater Fishes of South Australia (2 ed.). Adelaide, Australia: South Australian Government. - Hickman, C. P. Jr., Roberts, L. S., & Larson, A. (2002, June). Animal Diversity.
- Kendrick, A. J. & Hyndes, G. A. (2005). Variations in the dietary compositions of morphologically diverse syngnathid fishes. Environmental Biology of Fishes, 72, 415-427.
- Kuiter, R. H. 1980. Coastal Fishes of Southeastern Australia. Sydney, Australia: Gary Allen.
- Martin-Smith, K. M. (2011). Photo-identification of individual weedy seadragons Phyllopteryx taeniolatus and its application in estimating population dynamics. Journal of Fish Biology, 78(6), 1757-1768.
- Martin-Smith, K. M. & Vincent, A. C. J. (2006, April). Exploitation and trade of Australian seahorses, pipehorses, sea dragons and pipefishes (Family Syngnathidae). Oryx, 40(2), 141-151.
- MESA. (2015). Marine emblems. Marine Education Society of Astralasia (MESA).
- McGrouther, M. (2019, January 22). Common Seadragon, Phyllopteryx taeniolatus (Lacépède, 1804). Australian Museum.
- Morrison, S. & Storrie, A. (1999). Wonders of Western Waters: The Marine Life of South-Western Australia (1 ed.). Western Australia Department of Conservation and Land Management (CALM).
- Pogonoski, J. J., Pollard, D. A. and Paxton, J. R. (2002, February). Conservation Overview and Action Plan for Australian Threatened and Potentially Threatened Marine and Estuarine Fishes. Sydney, Australia: Environment Australia, Canberra.
- Pollom, R. (2017). Phyllopteryx taeniolatus. The IUCN Red List of Threatened Species 2017: e.T17177A67624517. http://dx.doi.org/10.2305/IUCN.UK.2017-2.RLTS.T17177A67624517.en
- Sanchez-Camara, J. & Booth, D. J. (2004). Movement, home range and site fidelity of the weedy seadragon Phyllopteryx taeniolatus (Teleostei: Syngnathidae). Environmental Biology of Fishes, 70, 31–41.
- Sanchez-Camara, J., Booth, D. J., & Turon, X. (2005). Reproductive cycle and growth of Phyllopteryx taeniolatus. Journal of Fish Biology, 67(1), 133-148.
- Sanchez-Camara, J., Booth, D. J., Murdoch, J., Watts, D., & Turon, X. (2006). Density, habitat use and
behaviour of the weedy seadragon Phyllopteryx taeniolatus (Teleostei : Syngnathidae) around Sydney, New South Wales, Australia. Marine and Freshwater Research, 57(7), 737-745. - Sanchez-Camara, J., Martin-Smith, K. M., Booth, D. J., Fritschi, J., & Turon, X. (2011). Demographics and
vulnerability of a unique Australian fish, the weedy seadragon Phyllopteryx taeniolatus. Marine Ecology
Progress Series, 422, 253-264. - Sea Life Melbourne. (2005, February 18). Weedy seadragon. Merlin Entertainments.
- Short, F. T. & Wyllie-Echeverria, S. (1996). Natural and human-induced disturbances of seagrasses. Environmental Conservation, 23: 17-27.
- The State of Victoria. (2011, November 21). The marine faunal emblem for the state of Victoria. Department of Sustainability and Environment.
- Stiller, J., Wilson, N. G., & Rouse, G. W. (2015, February 18). A spectacular new species of seadragon (Syngnathidae). Royal Society Open Science, 2(2), 1-12.
- Underwater Times New Service. (2012, August 4). Weedy sea dragons born at Monterey Bay Aquarium; Only 5th aquarium in North America with successful births. Underwater Times.
- The Wikimedia Foundation. (2019, March 14). Common Seadragon. Wikipedia.
- Wilson, N. G. & Rouse, G. W. (2010). Convergent camouflage and the non-monophyly of ‘seadragons’ (Syngnathidae:Teleostei): Suggestions for a revised taxonomy of syngnathids. Zoologica Scripta, 39, 551-558.