PHYSIOLOGY | IMAGES | ETYMOLOGY | TAXONOMY | GEOGRAPHY | BEHAVIOR | DIET | REPRODUCTION | ECOLOGY | CONSERVATION | FAUNAFACTS | VIDEO | SOURCES

The tiger shark is one of the largest ocean predators and relies on camouflage and stealth to capture its prey. This solitary, nocturnal carnivore is nomadic and can be found in the epipelagic and mesopelagic zones of neritic and oceanic habitats. The only viviparous species of its family, it gives birth to live young.
Physiology
Tiger sharks are large with a sizeable, blunt nose on the end of a wedge-shaped head. They are one of the largest predators in the ocean. Adults range from 3.25 to 4.25 meters in total length, although tiger sharks of 6 to 7.5 meters (25 feet) in length have been documented. Individuals larger than 5 meters, or 16 feet, in total length are rarely seen.
Tiger sharks display sexual dimorphism in body size as females are smaller than males. Female tiger sharks average 2.92 meters in total length while males average 3.20 meters in total length.
Adult tiger sharks typically weigh 385 to 635 kilograms, with the largest sharks reaching 862 kilograms.
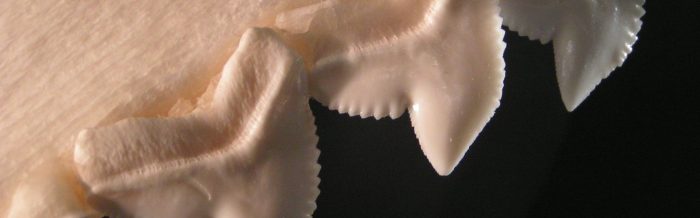
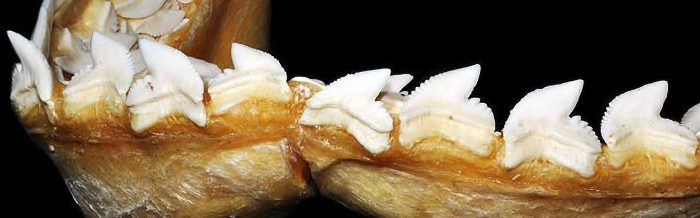
Tiger sharks have serrated teeth, making it easy to tear flesh and crack the bones and shells of their prey. Serrated teeth give this species the ability to penetrate the shells of sea turtles.
Tiger sharks have a heterocercal tail, meaning the dorsal lobe of the caudal fin is longer than the ventral lobe. In tiger sharks, the heterocercal tail, or caudal fin, is the primary source of propulsion. The caudal fin produces a downward thrust of water behind the center of balance in a shark, which should cause its head to turn upwards. However, because the tail also moves side to side, it keeps the head from turning upwards. Because of this, tiger sharks move in an S-shaped fashion.
Tiger sharks live longer in the wild than in captivity, averaging 27 years. The maximum age of a tiger shark is estimated at 27-37 years. Tiger sharks in captivity do not live as long, living a maximum of 17 to 20 years. In captivity, this species tends to die of starvation rather than old age, as food that is already dead is less appealing to tiger sharks.
Based on data from the Western North Atlantic and Australia, the generation length is estimated at 17.5-22.5 years.

SEXUAL DIMORPHISM
Larger MalesBODY LENGTH
3.25-7.5 m. / 10.66-24.6 ft.BODY MASS
385-862 kg. / 849-1,900 lb.LIFESPAN
15-37 yr.GENERATION LENGTH
17.5-22.5 yr.

Images
Taxonomy
The tiger shark was first described by Peron and Lesueur in 1822 and was given the scientific name Squalus cuvier. In 1837, Müller and Henle renamed it Galeocerdo tigrinus.
The tiger shark is the largest member of the Carcharhinidae family, commonly referred to as requiem sharks.
Carcharhiniformes is the most species-rich order of sharks with more than 270 species including the small catsharks and hammerhead sharks. Members of this order are characterized by the presence of a nictitating membrane over the eyes, two dorsal fins, an anal fin, and five gill slits. This family consists of mostly slender but powerful mid- to large-sized sharks and includes some other well-known sharks, such as the blue shark (Prionace glauca), lemon shark (Negaprion brevirostris), and bull shark (Carcharhinus leucas).
The tiger shark is the only extant member of the genus Galeocerdo.
KINGDOM
AnimaliaPHYLUM
ChordataCLASS
ChondrichthyesORDER
CarcharhiniformesFAMILY
CarcharhinidaeGENUS
GaleocerdoSPECIES
cuvierSUBSPECIES
None
Etymology
The tiger shark’s genus, Galeocerdo, is derived from the Greek galeos, which means shark, and kerdo, the word for fox. The tiger shark is often colloquially called the man-eater shark.
The tiger shark’s common name derives from the unique camouflage patterning of juveniles. Tiger shark pups are born with tiger-like stripes on their back and a lightly colored yellow or white belly. This camouflage patterning and coloration provides protection against predators and allowed the juveniles to to blend in with the environment. These stripes fade as the juveniles reach adulthood, which is around 6 to 8 years.
ALTERNATE
Leopard Shark, Man-Eater SharkGROUP
Frenzy, Gam, Herd, School, Shiver, ShoalYOUNG
Pup
Region
The tiger shark has a circumglobal range in subtropical and tropical, warm, temperate oceans, ranging between latitudes of approximately 40°N to 36°S.
In the western Atlantic, the species ranges from the state of Massachusetts in the United States of America to Uruguay, including the Gulf of Mexico, Latin America, islands of the Caribbean, and isolated islands such as Fernando de Noronha.
Tiger sharks have been sighted from the eastern coast of North America to the eastern coast of Brazil. In the eastern Atlantic, it is found along the West Africa coast, from Morocco to Angola, including the Canary Islands and the Azores.
It has been reported off Libya in the Mediterranean, though whether it is resident or vagrant is unknown.
This shark is distributed throughout the Indo-Pacific. In the Indian Ocean, the tiger shark is found from the east coast of Africa to South Africa, including remote islands such as Reunion and Chagos Archipelago, north to the Red Sea, and throughout the tropical Indian Ocean.
In the western and central Pacific, it occurs from Australia north to eastern China, India, and Japan, and throughout Pacific islands including Palau, east to the Solomon, Marshall, and Hawaiian Islands, south to northern New Zealand, as well as French Polynesia and more isolated atolls. In the eastern Pacific, it ranges from southern California to Peru, including the Galapagos and Revillagigedo Islands.
The tiger shark also appears seasonally in cool temperate waters, most likely following warmer currents. The presence of this shark in temperate regions has been reported from the United Kingdom and Iceland in the Atlantic, Alaska in the Northeast Pacific, the southern coasts of New South Wales and Western Australia in Australia, and in South Africa.
EXTANT
American Samoa, Angola, Anguilla, Antigua and Barbuda, Aruba, Australia, Bahrain, Barbados, Belize, Benin, Bermuda, Brazil, Cameroon, Cayman Islands, China, Colombia, Congo, Cook Islands, Costa Rica, Cuba, Côte d’Ivoire, Djibouti, Dominica, Dominican Republic, Ecuador, Egypt, Equatorial Guinea, Eritrea, Fiji, French Guiana, French Polynesia, Gabon, Gambia, Ghana, Grenada, Guadeloupe, Guam, Guatemala, Guinea, Guinea-Bissau, Guyana, Haiti, Honduras, India, Indonesia, Iran, Iraq, Jamaica, Kenya, Kiribati, Kuwait, Liberia, Maldives, Marshall Islands, Martinique, Mauritania, Mexico, Micronesia, Montserrat, Morocco, Mozambique, Nauru, New Caledonia, New Zealand, Nicaragua, Nigeria, Niue, Norfolk Island, Northern Mariana Islands, Oman, Pakistan, Palau, Panama, Papua New Guinea, Peru, Pitcairn, Portugal (Azores), Puerto Rico, Qatar, Saint Kitts and Nevis, Saint Lucia, Saint Vincent and the Grenadines, Samoa, Saudi Arabia, Senegal, Sierra Leone, Solomon Islands, Somalia, Sri Lanka, Sudan, Suriname, Taiwan, Tanzania, Timor-Leste, Togo, Tokelau, Tonga, Trinidad and Tobago, Tuvalu, United Arab Emirates, United Kingdom, United States, Uruguay, Vanuatu, Venezuela, Vietnam, Virgin Islands, British, Wallis and Futuna, Western Sahara, Yemen (Socotra)VAGRANT
Iceland, Libya, South Africa, United States (Alaska)
Habitat
The tiger shark is a saltwater species and inhabits marine neritic and oceanic habitats. It can be found near shelf, reef, and slope habitats and is sometimes associated with coral reefs. Although it prefers the sea grass ecosystems of the coastal areas, it occasionally inhabits other areas due to prey availability.
Tiger sharks have also been documented in river estuaries and harbors.
The tiger shark is found in the epipelagic zone of 0-200 meters and occasionally makes longer-distance excursions into the mesopelagic zone of 200-1,000 meters. Tiger sharks spend approximately 36% of their time in shallow coastline habitats, generally at depths of 2.5 to 145 meters. The species mostly occurs to 100-meter depth but has also been documented several kilometers from the shallow areas and at depths greater than 1,000 meters with 1,136 meters being the maximum recorded.
Female tiger sharks are observed in shallow areas more often than males.
The tiger shark is highly mobile and has been noted to travel hundreds, and in some cases thousands, of kilometers.
MARINE NERITIC
Pelagic, Subtidal Rock and Rocky Reefs, Subtidal Sandy, Coral Reef, Submerged Seagrass, EstuariesMARINE OCEANIC
Epipelagic, Mesopelagic
Co-Habitants
Behavior
Tiger sharks are solitary except during the mating seasons or while communally feeding on large carcasses.
The tiger shark is nomadic and highly mobile. This species has been noted to travel hundreds, and in some cases thousands, of kilometers. Individuals attached with transmitters swam up to 16 kilometers in a single day and did not return to that area for close to a year. As a result, tiger sharks have very large home ranges and territories of about 23 square kilometers.
The tiger shark relies on electromagnetic receptors to perceive its environment and to hunt prey. Sensing organs called Ampullae of Lorenzini, located on the end of their nose, are filled with a jelly-like substance that reads electromagnetic signals. These signals are sent from the pores to the sensory nerve, and then to the brain. While hunting, tiger sharks use this ability to detect electromagnetic signals given off by fish. Tiger sharks also use these organs to sense changes in water pressure and temperature. Members of this species also have a lateral line on both sides of the body that runs from the gill line to the base of the tail. The lateral line reads the vibrations in the water from the movement of other animals nearby. Ampullae of Lorenzini and lateral lines also help detect electromagnetic signals from other sharks. While communally feeding on carcasses, sharks give off signals signifying dominance and thus the order in which they feed.
CIRCADIAN RHYTHM
Nocturnal
Diet
The diet of tiger sharks includes mollusks, birds, snakes, crustaceans, sea turtles, and dugongs (Dugong dugon). Tiger sharks also often scavenge dead or injured whales.
As top predators in their ecosystem, it is possible that tiger sharks control populations of prey species, although this has not been verified.
Tiger sharks are nocturnal predators and most likely rely on stealth rather than strength and speed to catch prey. Tiger sharks are well camouflaged, allowing them to get within striking range of prey. They are blue or green in color with a light yellow or white under-belly. If prey flee, tiger sharks may back off, not taking part in high-speed pursuits. However, tiger sharks are capable of short bursts of speed once their prey are within range. They are very strong and fast and are not quickly or easily exhausted.
While communally feeding on carcasses, sharks give off signals signifying dominance and thus the order in which they feed. During these group feedings, tiger sharks have a loose social hierarchy where larger sharks feed first. Smaller sharks circle around the carcass until the larger sharks are full, then move in to feed. Violence is minimal during these scavenging feasts.
Large tiger sharks can survive several weeks without feeding.
PREFERENCE
GeneralistSTYLE
Ambush
Prey

Dugong
Reproduction
Tiger sharks are polygynandrous, meaning males and females have multiple mates. Tiger sharks do not pair-bond at any time. Not much is known regarding the behaviors of finding, attracting, and defending mates.
There is regional variability in tiger shark sizes and ages at sexual maturity. On average, tiger sharks reach sexual maturity at 4-13 years of age. Male tiger sharks reach maturity earlier than females, maturing when they reach an average total length of 25 to 30.5 meters, whereas females reach sexual maturity when they reach 27.4 to 34.5 meters in length.
Female tiger sharks mate once every three years.
Tiger shark breeding seasons differ in the northern and southern hemispheres. In the northern hemisphere, females delay fertilization until March or May in order to give birth between May and June of the following year. In the southern hemisphere, females delay mating until November or January in order to give birth between February and March of the following year.
Male tiger sharks have diametric testes, which are capable of synthesizing a larger amount of sperm than radial or compound testes. The females have external ovaries that appear on the epigonal organ, which is a primary lymphoid tissue in elasmobranchs.
The tiger shark is the only species of the family Carcharhinidae that is lecithotrophic viviparous, giving birth to live young. Female tiger sharks typically give birth in a nursery, which provides protection during the birthing process and continues to protect the young directly after birth in the absence of parents.
Gestation is approximately 13-16 months with a triennial reproductive cycle in Hawaii and Australia.
Embryos of tiger sharks are fertilized internally and a yolk sac forms around them to provide necessary nutrients during the gestation period. As the yolk begins to run out near the end of the gestation, the embryo draws nutrients directly from the mother.
The tiger shark has a higher intrinsic rate of population increase than many other carcharhinid sharks. Litter sizes are relatively large, ranging from 10-82 pups. With a maximum of 82 embryos recorded, an average tiger shark litter consists of 26-33 pups. Many of these pups will not survive to adulthood.
Tiger sharks have no parental investment as mothers do not help their pups survive or find food or shelter. Male tiger sharks also play no role in the lives of their offspring.
Instead, tiger shark pups are born fully developed and independent with traits that help them survive without parents. This includes camouflage patterning and coloration to provide protection against predators, teeth to help capture prey, and speed to avoid predators. Tiger sharks also have relatively fast growth rates during early life stages. At birth, they range from 51-90 centimeters in total length and weigh 3 to 6 kilograms.
Tiger shark pups are born with tiger-like stripes on their back and a lightly colored yellow or white belly which allows them to blend in with the environment. These stripes fade as the juveniles reach adulthood, which is around 6 to 8 years.
BREEDING SEASON
November-MayBREEDING INTERVAL
3 YearsPARENTAL INVESTMENT
NoneGESTATION
13-16 MonthsBIRTHING SEASON
February-JuneLITTER
10-82INDEPENDENCE
BirthSEXUAL MATURITY
4-13 Years
Ecology
Tiger sharks are some of the largest predators in the ocean and have few species that feed on them. Some juvenile tiger sharks, however, fall prey to other sharks. Humans also fish for tiger sharks.
Tiger sharks serve as a host for remoras, which are small suckerfish. Tiger sharks and remoras share a commensal relationship as the remoras attach to the tiger sharks near the underbelly and use the sharks for transportation and protection. Remoras also feed on materials dropped by tiger sharks. Recently, copepods, specifically sea louse (Caligus oculicola), have also been discovered around the eyes of tiger sharks in Australia.
Although very unlikely, tiger sharks enter shallow, populated areas of coastlines and attack humans on rare occasions.
Tiger sharks are a popular gamefish, which are typically captured and released for sport. In some U.S. states, permits such as a saltwater fishing license, allow fishermen to collect the shark as a trophy. Fishing for these sharks is tiring as tiger sharks are very fast, strong, and not quickly or easily exhausted. Tiger sharks often perform aerial acts when hooked.
Globally, the tiger shark is caught by commercial, recreational, small-scale, and artisanal fisheries as a target species or incidental bycatch and is subject to illegal, unreported, and unregulated (IUU) fishing operations. The species is one of the major components in catches from shark control programs that target large shark species in Australia, South Africa, and more recently Reunion Island in the Western Indian Ocean.
The tiger shark is valued for its high quality fins, skin, liver oil, meat, and cartilage. The tiger shark has been increasingly exploited by fisheries since the 1950’s due to the increasing demand from the shark fin trade as the fins of this species are a common component of the Hong Kong fin trade.
Catches of tiger shark in directed shark fisheries have been documented for a number of regions including the western Atlantic, Brazil, Australia, Papua New Guinea, Peru, Taiwan, India, and Saudi Arabia.
The tiger shark is a common component of the U.S. east coast/Gulf of Mexico commercial shark bottom longline fishery, accounting for 8-36% of the catch. The fishery catches mostly juvenile and sub-adult tiger sharks, although some larger sharks are also taken.
In Australia, the tiger shark is targeted by commercial shark fisheries in northern New South Wales and in Western Australia. The species accounted for 3 tonne (t) and 5.9% of total catch of the Ocean Trap and Line Fishery in eastern Australia. However, catches by this fishery are probably smaller than landings from recreational fisheries in that same area. In the Western Australia Tropical Shark Fishery, this shark was caught as a target species and annual catches averaged approximately 41 t between 2000 and 2004. This fishery was closed in late 2005. The Offshore Net and Line Fishery operating in the northern Australia catches the species incidentally (27 t in 2012), although mesh size is assumed to prevent capture of greater numbers. This shark is also taken in the Southern and Western Demersal Gillnet and Demersal Longline Fishery in Western Australia with a catch of 112 t reported in 2005-06. The Tiger Shark is a bycatch in trawl fisheries although normally in small numbers and there are few records of catches for these fisheries. The Australian Commonwealth Trawl Sector reported a total catch for the species of 4.7 t from 2004-2011.
The tiger shark is relatively common in the Indonesian shark fishery and contributed 5.2% of the total shark biomass of catches from between 2001 and 2006. The tiger shark is also commonly caught in the seamount gillnet and longline fishery off the west coast of India, where 242 t and 144 t of various shark species were taken during 2010 and 2011, respectively, but species-specific catches were not recorded. The Tiger Shark is caught occasionally in the longline fisheries of Costa Rica, Mozambique, and Saudi Arabia and by purse seiners in the Indian Ocean. Although little species-specific catch data information is available for the Arabian Seas area, the high level of level of fishing pressure in the region is of concern with steep increases in both small-scale and industrial fishing effort between the 1990s and 2000s.
In tuna longline fisheries, the species is typically caught in small numbers relative to pelagic sharks and is often not reported. Between 2007 and 2013, an average of 54 t was landed annually by tuna longline boats regulated by the International Commission for the Conservation of Atlantic Tuna. Tiger shark represented less than 10% of the total bycatch of all shark species in each of the management zones of the U.S. pelagic longline fishery. In the Panama longline fishery, the tiger shark represented 1.6% of shark catches of the estimated total catch of 109,500 t of various sharks since the mid-1980’s. In the Central and Western Pacific, the tiger shark is caught in low numbers (0.025 sharks per set) by tuna longline boats from most countries operating in that region. The tiger shark is a common target of recreational fisheries in the United States, Australia, and South Africa. Approximately 96% of the catch in USA waters are released alive. In the New South Wales Gamefish Tournament, tiger shark catches were approximately 8 t per year between 1993 and 2005. Recreational fishing may account for mortality in the tiger shark population in other countries, although catches are unmonitored.
Artisanal fisheries and IUU fisheries are also likely to be catching tiger shark, however information about landings from these fisheries is scarce as they remain mostly unmonitored. The tiger shark is commonly caught in artisanal fisheries in the tropics and subtropics, including in Mexico, Panama, Brazil, and a few African countries. In Bangladesh, landings of 4.5 t represent on average 1.36% of total catch. There is evidence that the species has been overfished by Indonesian fishing boats at Ashmore and Cartier Islands, and Scott Reef in northern Australian waters. This area had been targeted by Indonesian fishermen since the 1800’s but fishing was banned in 1988 and 2000, respectively. A survey in the region showed the absence of tiger shark at reefs historically fished despite years of protection whereas they were found present at nearby atolls that had been always protected from fishing. The species comprised 19% of the total shark biomass and 7.4% of total catch in numbers from Indonesian and Taiwanese IUU fishing vessels.
The tiger shark is a target species of shark control programs in Queensland and New South Wales, Australia, in South Africa, and more recently at Reunion Island. In Australia, the Queensland Shark Control Program (QSCP) captured 4,757 tiger shark individuals between 1993 and 2010 and the species represented approximately 10-30% of total catch in the northern locations of the QSCP between 1964 and 2007. Standardized catch rates from 1964-2007 for the QSCP in northern Queensland showed an increase in the relative importance of the tiger shark, from approximately 10 to 30% of total catch. This pattern is most likely due to the move in fishing gear from nets to drumlines. In southeast Australia, the tiger shark was commonly captured by the New South Wales Shark Meshing Program from 1950-2010 with approximately 30 sharks per year between 1950 and 2008, representing approximately 10% of the common shark species caught by the program in each of its locations. In South Africa, the KwaZulu-Natal beach protection program has caught approximately 50 individuals annually from 1978-2003. Since 1989, sharks caught alive are released; however, fishing mortality of the tiger shark in nets is 27% of captures.
SPORT HUNTING/SPECIMEN COLLECTING
Local, National, InternationalWEARING APPAREL, ACCESSORIES
Local, National, InternationalFOOD
Local, National, International
Predators

Human
Conservation
In the Arabian Seas region, the tiger shark has been assessed as Vulnerable based on a suspected decline of at least 30% over the past three generations based on levels of exploitation, with the continued demand from the shark fin market suspected to result in further population reduction in the future.
Globally, the tiger shark is suspected to have declined by close to 30% over the past three generations (53-68 years) due to exploitation from commercial, recreational, and unregulated fisheries, as well as shark control programs. A further population reduction is suspected over the future three generation lengths (2018-2086) based on current levels of exploitation that are likely to continue into the future.
As such this species is assessed as Near Threatened and is close to meeting Vulnerable A2bd+3d.
Population
In Australia, the Queensland Shark Control Program showed declines in catch rates since the 1980’s and in New South Wales, evidence of a decline was also seen in the last 20 years. Additionally, a significant reduction of the proportion of mature sharks in catches over 60 years suggests shifts in population structure and raises concerns over possible effects from this program on the Tiger Shark population off New South Wales. The decline off eastern Australia is likely to continue while the shark control programs off Queensland and New South Wales remain active.
In the U.S. fisheries of the Western North Atlantic, observer surveys variously recorded increases in tiger shark relative abundance and no change in abundance. In South Africa, the beach protection program showed a 3% increase per year in tiger shark catch rates. The increasing rates in the Western North Atlantic and South Africa seem consistent with the pattern observed in the first 20 years in Queensland and the interpretation of such upward trends should be made with caution.
Currently, the total number of tiger sharks worldwide is unknown.
Given the wide range of the species and the localities of the shark control programs, the tiger shark is suspected to have undergone an overall population reduction of close to 30% globally over the past three generations (53- 68 years) based on actual levels of exploitation from commercial, recreational, and unregulated fisheries, as well as shark control programs.
A further population reduction is suspected over the future three generation lengths (2018-2086) based on current levels of exploitation that are likely to continue into the future.
THREATS
The tiger shark has relatively fast growth rates and large litters, but the likely triennial reproductive cycle reduces its ability to recover from fishing pressure. The tiger shark has been shown to be at risk in the Arabian Seas region from fishing pressure and from some shark control programs in other regions. The widespread distribution of this species increases the likelihood that it will survive increasing levels of exploitation in certain areas.
Records of tiger shark catches by many fisheries globally are still largely unknown. The species is not commonly caught in large numbers by most commercial fisheries and its catch is rarely recorded. Long-term trends of tiger shark catch rates are available from regional shark control programs and United States fisheries observer programs. Available trends in catch rates are based on analyses of the long-term shark control programs and on observer surveys from the Western North Atlantic. Consequently, there is a lack of adequate data for stock assessment, definition of population structure, and documentation of overall global population trends for the tiger shark.
In the Western North Atlantic, data from observer surveys taken between 1960 and 2000 from the Northeast Fisheries Science Center showed an upward trend of the relative abundance of the tiger shark with an increase of 6.7% per year since the 1980’s. Catch-per-unit-effort (CPUE) logbook data indicated a slow decline from 1986 to 2000 in tiger shark relative abundance. When logbook and observer data were combined, the relative abundance declined slightly then increased. The Shark Bottom Longline Observer Program also showed an increase of 8% per year in tiger shark relative abundance between 1994 and 2013. However, the Pelagic Observer Program from the Southeast Fisheries Science Center showed no change in abundance for the same period.
In northeast Australia, the Queensland Shark Control Program (QSCP) has used a combination of nets and drumlines to target large sharks since 1962. The standardized catch rate of tiger
sharks in northern Queensland increased until the 1980’s and then decreased by the 2000’s to approximately 66% of its original value. This trend in catch rate suggests a possible decline in the population between 1964 and 2007. The CPUE of tiger sharks showed an overall declining trend for the QSCP between 1993 and 2010, with the most significant decline in southern Queensland.
In southeast Australia, New South Wales has operated a netting program since 1937. The CPUE of tiger sharks showed marked fluctuations in the first four decades of records, and did not significantly change between 1950 and 2010, although a downward trend in CPUE was observed in the last two decades. The size frequency of tiger sharks caught by the program has changed over 60 years with a significant reduction in the proportion of large mature individuals being caught. The recent decline in catch rate and decrease in the proportion of large individuals suggests changes in the population structure and raises concern over the effects the program is having on the tiger shark population off New South Wales. The New South Wales
game fishing tournaments reported a decline in CPUE from approximately 1.2 to 0.5 shark vessel per day between 1993/94 and 2004/05, with fishers still targeting tiger sharks during the tournaments, suggesting a decline in the population.
The decline off eastern Australia is likely to continue while the shark control programs off Queensland and New South Wales remain active.
In South Africa, the KwaZulu-Natal beach protection program has deployed large-mesh gillnets since 1952. There has been an annual increase of 3% in the CPUE of tiger sharks between 1978 and 2003. This pattern seems comparable to the pattern observed in the first two decades of the Queensland Shark Control Program in northern Queensland, but the interpretation of such upward trends should be made with caution.
In the Arabian Seas, a significant decrease in shark biomass, mainly carcharhinids, has been reported since the 1970’s from trawl surveys and interviews with fishers. The tiger shark was formerly common in Eritrea and Iranian Gulf waters, but interviews with fishermen have indicated large declines in landings. Surveys in the UAE have recorded a decline of approximately 90% in the landings of the tiger shark and the species has been absent from recent surveys in the Red Sea, where once it was reported to be common. In Pakistan and India, declines of 20-30% in the tiger shark have been recorded with an absence of individuals larger than 3 meters in length in catches. The tiger shark is suspected to have undergone declines of at least 30-50% in the Arabian Sea over the last three generations (53 years) based on current levels of exploitation. The tiger shark was assessed as Vulnerable A2cd+3cd in the Arabian Seas region and the decline is likely to be ongoing with the continued demand from the shark fin market suspected to result in further population reduction in the future.
BIOLOGICAL RESOURCE USE
Fishing & Harvesting Aquatic Resources
ACTIONS
There are no specific conservation or management measures in place for the tiger shark. However, there has been an increasing interest to intensify protection of the species in the Western North Atlantic.
The current Fisheries Management Program for the U.S. Atlantic and Gulf of Mexico includes quotas and fishing seasons to regulate catch of coastal shark species including the tiger shark. On the west and south coasts of Western Australia, a maximum size of 70 centimeters interdorsal length is applied for whaler sharks taken by recreational shark fishing, which includes the tiger shark. In Queensland, the maximum size allowed is 150 centimeters for all shark species and a possession limit of one shark of any species is imposed to recreational fishing. Similarly, in New South Wales, only one tiger shark can be taken per person per day.
Some countries in the Arabian Seas region, such as Kuwait, Saudi Arabia, Sudan, and the Maldives, have banned the targeted fishing for sharks, whereas others, such as Iran and the UAE, impose seasonal bans on shark fishing.
FaunaFacts
Video
SourceS
- Afonso, A. S. & Hazin, F. H. V. (2015). Vertical movement patterns and ontogenetic niche expansion in the tiger shark, Galeocerdo cuvier. Public Library of Science (Plos) One, 10(1), e0116720.
- Afonso, A. S., Hazin, F. H. V., Barreto, R. R., Santana, F. M. & Lessa, R. P. (2012). Extraordinary growth in tiger sharks Galeocerdo cuvier from the South Atlantic Ocean. Journal of Fish Biology, 81, 2080-2085.
- Akhilesh, K. V., Ganga, U., Pillai, N. G. K., Vivekanandan, E., Bineesh, K. K., Shanis, C. P. R., & Hashim, M. (2011). Deep-sea fishing for chondrichthyan resources and sustainability concerns— a case study from southwest coast of India. Indian Journal of Geo-Marine Sciences, 40(3), 347-355.
- Baum, J. K. & Blanchard, W. (2010). Inferring shark population trends from generalized linear mixed models of pelagic longline catch and effort data. Fisheries Research, 102, 229-239.
- Baum, J. K., Myers, R. A., Kehler, D. G., Worm, B., Harley, S. J., & Doherty, P. A. (2003, February). Collapse and conservation of shark populations in the Northwest Atlantic. Science, 299, 389-392.
- Berkeley, S. A. & Campos, W. L. (1988). Relative abundance and fishery potential of pelagic sharks along Florida’s east coast. Marine Fisheries Review, 50(1), 9–16.
- Bineesh, K. K., Akhilesh, K. V., Abdussamad, E. M., & Prakasan, D. (2014). Seamount associated fishery of south-west coast of India: A preliminary assessment. Indian Journal of Fisheries, 61(3), 29-34.
- Bonfil, R. (1994). Overview of world elasmobranch fisheries. FAO Fisheries Technical Paper. Rome, Italy: Food and Agriculture Organization of the United Nations Rome (FAO).
- Bornatowski, H., Braga, R. R., Abilhoa, V., & Corrêa, M. F. M. (2014). Feeding ecology and trophic
- comparisons of six shark species in a coastal ecosystem off southern Brazil. Journal of Fish Biology, 85, 246-263.
- Branstetter, S., Musick, J. A., & Colvocoresses, J. A. (1987, February). A comparison of the age and growth of the tiger shark, Galeocerdo cuvieri, from off Virginia and from the northwestern Gulf of Mexico. NMFS Fishery Bulletin, 85(2), 269–79.
- Bruckner, A. W., Alnazry, H. H., & Faisal, M. (2011). A paradigm shift for fisheries management to enhance recovery, resilience, and sustainability of coral reef ecosystems in the Red Sea. In: W. W. Taylor, A. J. Lynch, & M. G. Schechter (Eds.), Sustainable Fisheries: Multi-Level Approaches to a Global Problem (pp. 85–111). Herndon, VA: American Fisheries Society.
- Carlson, J. K., Hale, L. F., Morgan, A., & Burgess, G. (2012). Relative abundance and size of coastal sharks derived from commercial shark longline catch and effort data. Journal of Fish Biology, 80, 1749-1764.
- Chassot, E., Floch, L., Dewals, P., Damiano, A., Cauquil, P., & Chavance, P. (2014, November 3). Fishing activities of the French and associated flags purse seiners targeting tropical tunas in the Indian Ocean (1981-2013). IOTC-2014-WPTT16-XX.
- Clarke, S. C., McAllister, M. K., Milner-Gulland, E. J., Kirkwood, G. P., Michielsens, C. G. J., Agnew, D. J., Pikitch, E. K., Nakano, H., & Shivji, M. S. (2006). Global estimates of shark catches using trade records
- from commercial markets. Ecology Letters, 9, 1115-1126.
- Cliff, G. & Dudley, S. F. J. (2011). Reducing the environmental impact of shark-control programs: A case study from KwaZulu-Natal, South Africa. Marine and Freshwater Research, 62, 700-709.
- Compagno, L. J. V. (2002). Sharks of the World: An Annotated and Illustrated Catalogue of Shark Species to
- Date. (Vol. 2). Rome, Italy: Food and Agriculture Organization of the United Nations Rome (FAO) Fisheries Synopsis.
- Cressey, R. F. & Lachner, E. A. (1970). The parasitic copepod diet and life history of diskfishes (Echeneidae). Copeia, 1970(2), 310-318.
- Dapp, D., Arauz, R., Spotila, J. R., & O’Connor, M. P. (2013). Impact of Costa Rican longline fishery on its bycatch of sharks, stingrays, bony fish and olive ridley turtles (Lepidochelys olivacea). Journal of Experimental Marine Biology and Ecology, 448, 228-239.
- Department of Fisheries. (2005, November). >em>Application to the Australian Government Department of Environment and Heritage on the Western Australian Tropical Shark Fisheries (Covering the Western Australia North Coast Shark Fishery and the Joint Authority Northern Shark Fishery). Cloisters Square, WA: Department of Fisheries, Government of Western Australia.
- Dicken, M. L. & Hosking, S. G. (2009). Socio-economic aspects of the tiger shark diving industry within the Aliwal Shoal Marine Protected Area, South Africa. African Journal of Marine Science, 31, 227-232.
- Dill, L., Heithaus, M., & Walters, C. (2003). Behaviorally mediated indirect interactions in marine communities and their conservation implications. Ecology, 84(5), 1151-1157.
- Draper, K. (2011). Galeocerdo cuvier. Animal Diversity Web.
- Driggers III, W. B., Ingram, G. W., Grace, M. A., Gledhill, C. T., Henwood, T. Horton, C. & Jones, C. M. (2008). Pupping areas and mortality rates of young tiger sharks Galeocerdo cuvier in the western North Atlantic Ocean. Aquatic Biology, 2: 161-170.
- Dudley, S. F. J. & Simpfendorfer, C. A. (2006). Population status of 14 shark species caught in the protective gillnets off KwaZulu-Natal beaches, South Africa, 1978-2003. Marine and Freshwater Research, 57, 225-240.
- Ebert, D. A., Fowler, S., & Compagno, L. (2013). Sharks of the World: A Fully Illustrated Guide. Plymouth, United Kingdom: Wild Nature Press.
- Essapian, F. (1962). Notes on the behavior of sharks in captivity. Copeia, 1962(2), 457-459.
- Ferreira, L. C. & Simpfendorfer, C. (2019). Galeocerdo cuvier. The IUCN Red List of Threatened Species 2019: e.T39378A2913541. http://dx.doi.org/10.2305/IUCN.UK.2019-1.RLTS.T39378A2913541.en.
- Ferreira, L. C., Thums, M., Meeuwig, J. J., Vianna, G. M. S., Stevens, J., McAuley, R., & Meekan, M. G. (2015). Crossing latitudes—long-distance tracking of an apex predator. Public Library of Science (Plos) One, 10(2), e011691.
- Field, I. C., Meekan, M. G., Buckworth, R. C., & Bradshaw, J. A. (2009). Protein mining the world’s oceans: Australasia as an example of illegal expansion-and-displacement fishing. Fish and Fisheries, 10, 323–328.
- Garcia, V., Lucifora, L., & Myers, R. (2008). The importance of habitat and life history to extinction risk in sharks, skates, rays and chimaeras. Proceedings of the Royal Society B: Biological Sciences, 275(1630), 83-89.
- Gonzalez-Pestana, A., Kouri J. C., & Velez-Zuazo, X. (2014). Shark fisheries in the Southeast Pacific: A 61-year analysis from Peru. F1000Research, 3(164), 1.
- Gruber, S., & Myrberg, Jr., A. (1977). Approaches to the study of the behavior of sharks. American Zoologist, 17(2), 471-486.
- Harper, S., Guzmán, H. M., Zylich, K., & Zeller, D. (2014). Reconstructing Panama’s total fisheries catches from 1950 to 2010: Highlighting data deficiencies and management needs. Marine Fisheries Review, 76,
- 51-65.
- Heithaus, M. (2001). The biology of tiger sharks, Galeocerdo Cuvier, in Shark Bay, Western Australia: Sex ratio, size distribution, diet, and seasonal changes in catch rates. Environmental Biology of Fishes, 61(1), 25-36.
- Heithaus, M., Dill, L., Marshall, G., & Buhleier, B. (2002). Habitat use and foraging behavior of tiger sharks (Galeocerdo cuvier) in a seagrass ecosystem. Marine Biology, 140(2), 237-248.
- Heithaus, M., Frid, A., Wirsing, A., Dill, L., Fourqurean, J., Burkholder, D., Thomson, J., & Bejder, L. (2007). State-dependent risk-taking by green sea turtles mediates top-down effects of tiger shark intimidation in a marine ecosystem. Journal of Animal Ecology, 76(5), 837–844.
- Heithaus, M., Hamilton, I., Wirsing, A., & Dill, L. (2006). Validation of a randomization procedure to assess animal habitat preferences: Microhabitat use of tiger sharks in a seagrass ecosystem. Journal of Animal Ecology, 75(3): 666-676.
- Hoey, J. J. & Casey, J. G. (1986). Shark Longline Fisheries: Gear and Production Characteristics. Proceedings of the Gulf & Caribbean Fisheries Institute.
- Holland, K., Wetherbee, B., Lowe, C., & Meyer, C. (1999). Movements of tiger sharks (Galeocerdo cuvier) in coastal Hawaiian waters. Marine Biology, 134(1), 665-673.
- Holmes, B. J. (2015). The biology and ecology of the tiger shark (Galeocerdo cuvier) on the east coast of Australia (Doctoral dissertation). Queensland, Australia: School of Biological Sciences, University of Queensland.
- Holmes, B. J., Peddemors, C. M., Gutteridge, A. N., Geraghty, P. T., Chan, R. W. K., Tibbetts, I. R., & Bennett, M. B. (2015). Age and growth of the tiger shark Galeocerdo cuvier off the east coast of Australia. Journal of Fish Biology, 87, 422-448
- Holmes, B. J., Pepperell, J. G., Griffiths, S. P., Jaine, F. R., Tibbetts, I. R., & Bennett, M. B. (2014). Tiger shark (Galeocerdo cuvier) movement patterns and habitat use determined by satellite tagging in eastern Australian waters. Marine Biology, 161, 2645-2658.
- Holmes, B. J., Sumpton, W. D., Mayer, D. G., Tibbetts, I. R., Neil, D. T., & Bennett, M. B. (2012). Declining trends in annual catch rates of the tiger shark (Galeocerdo cuvier) in Queensland, Australia. Fisheries Research, 129-130, 38-45.
- Jabado, R. W., Ghais S. M. A, Hamza, W., Henderson, A. C., Spaet, J. L. Y., Shivji, M. S., & Hanner, R. H. (2015). The trade in sharks and their products in the United Arab Emirates. Biological Conservation, 181, 190–198.
- Jabado, R. W., Kyne, P. M., Pollom, R. A., Ebert, D. A., Simpfendorfer, C. A., Ralph, G. M., & Dulvy, N. K. (2017). The Conservation Status of Sharks, Rays, and Chimaeras in the Arabian Sea and Adjacent Waters. Vancouver, Canada: Environment Agency – Abu Dhabi, UAE and IUCN Species Survival Commission Shark Specialist Group.
- Jabado, R. W. & Spaet, J. L. Y. (2017). Elasmobranch fisheries in the Arabian Seas Region: Characteristics, trade and management. Fish and Fisheries, 18, 1096–1118.
- Jit, R. B., Ali, M. H., Singha, N. K., Rahman, G., & Alam, F. (2014). Sharks and rays fisheries of the Bay of Bengal at the landing centers of Chittagong and Cox’s Bazar, Bangladesh. Global Journal in Science Frontiers Research, D 14, 79-89.
- Kalmijn, A. (2000). Detection and processing of electromagnetic and near-field acoustic signals in elasmobranch fishes. Philosophical Transactions: Biological Sciences, 355(1401), 1135-1141.
- Kattan, A. (2014, December). Baselines and Comparison of Coral Reef Fish Assemblages in the Central Red Sea (Master’s thesis). Thuwal, Kingdom of Saudi Arabia: King Abdullah University of Science and Technology.
- Kneebone, J., Natanson, L .J., Andrews, A. H., & Howell, W. H. (2008). Using bomb radiocarbon analyses to validate age and growth estimates for the tiger shark, Galeocerdo cuvier, in the western North Atlantic. Marine Biology, 60, 1-87.
- Kumoro, L. (2013). The Shark Longline Fishery in Papua New Guinea. Mooloolaba, Australia: 176th Meeting of the Standing Committee on Tuna and Billfish.
- Lea, J. S. E., Wetherbee, B. M., Queiroz, N., Burnie, N., Aming, C., Sousa, L. L., Mucientes, G. R., Humphries, N. E.,Harvey, G. M., Sims, S. W., & Shivji, M. S. (2015, July). Repeated, long-distance migrations by a philopatricpredator targeting highly contrasting ecosystems. Scientific reports, 5(11202).
- Lyle, J. M., Pyne, R. R., Hooper, J., & Croaker, S. L. (1984). North Australia’s multi-species shark fishery: A preparatory evaluation of the development of a shark fishing industry in northern territory waters. North Australia’s Multi-Species Shark Fishery, 1(12), 1-36.
- Macbeth, W. G., Geraghty, P. T., Peddemors, V. M., & Gray, C. A. (2009). Observer-based study of targeted commercial fishing for large shark species in waters off northern New South Wales. Cronulla, NSW: Industry & Investment NSW – Fisheries Final Report Series. Cronulla Fisheries Research Centre of Excellence.
- Mandelman, J. W., Cooper, P. W., Werner, T. B., & Lageux, K. M. (2008). Shark bycatch and depredation in the U.S. Atlantic pelagic longline fishery. Reviews in Fish Biology and Fisheries, 18, 427-442.
- Marshall, L. J. (2011). The Fin Blue Line: Quantifying Fishing Mortality Using Shark Fin Morphology. (Doctoral Dissertation). University of Tasmania, Hobart, Australia.
- Matsumoto, T., Saito, H., & Miyabe, N. (2004, January). Report of observer program for Japanese tuna longline fishery in the Atlantic Ocean from August 2003 to January 2004. Collect. Vol. Sci. Pap. ICCAT, 58, 1694- 1714.
- Mecklenburg, C. W., Mecklenburg, T. A., & Thorsteinson, L. K. (2002, June 1). Fishes of Alaska. Bethesda, MD: American Fisheries Society.
- Meekan, M. G., Vigliola, L., Hansen, A., Doherty, P. J., Halford, A., & Carleton, J. H. (2006). Bigger is better: Size-selective mortality throughout the life history of a fast-growing clupeid, Spratelloides gracilis. Marine Ecology Progress Series, 317, 237-244.
- Meyer, C. G., O’Malley, J. M., Papastamatiou, Y. P., Dale, J. J., Hutchinson, M. R., Anderson, J. M., Royer, M., & Holland, K. N. (2014). Growth and maximum size of tiger sharks (Galeocerdo cuvier) in Hawaii. Public Library of Science (Plos) One, 9, e84699.
- Morgan, A., Cooper, P. W., Curtis, T., & Burgess, G. H. (2009). Overview of the U.S. East coast bottom longline shark fishery, 1994-2003. Marine Fisheries Review, 71, 23-38.
- Natanson, L. J., Casey, J. G., Kohler, N. E., & Colket, T. (1999). Growth of the tiger shark, Galeocerdo cuvier, in the western North Atlantic based on tag returns and length frequencies; and a note on the effects of tagging. Fisheries Bulletin, 97, 944-953.
- Park, T. (2007). NSW Gamefish Tournament Monitoring: Angling Research Tournament Monitoring Program. Cronulla, NSW: NSW Department of Primary Industries.
- Paterson, R. A. (1990). Effects of long-term anti-shark measures on target and non-target species in Queensland, Australia. Biological Conservation, 52, 147–159.
- Pepperell, J. (1992). Trends in the distribution, species composition and size of sharks caught by gamefish
- anglers off south-eastern Australia, 1961-1990. In J. Pepperell (Ed.), Sharks: Biology and Fisheries (pp. 213-225). Melbourne, Australia: Australian Journal of Marine and Freshwater Research, CSIRO Publications.
- The Regional Organization for the Conservation of the Environment of the Red Sea and Gulf of Aden (PERSGA). (2003, March). Status of the Living Marine Resources in the Red Sea and Gulf of Aden and Their Management: Strategic Action Programme for the Red Sea and Gulf of Aden. Jeddah, Saudi Arabia: Regional Organization for the Conservation of the Environment of the Red Sea and Gulf of Aden.
- Pimenta, E. G., Vieira, Y. C., Rodrigues, T., & Amorim, A. F. (2014). Analysis of small-size fleet fishery based in Cabo Frio city, Rio de Janeiro-Brazil (2003-2012). Collect. Vol. Sci. Pap. ICCAT, 70(6), 2856-2867.
- Pratt, Jr., H. (1988). Elasmobranch gonad structure: A description and survey. Copeia, 1988(3), 719-729.
- Ramirez-Amaro, S. R., Cartamil, D., Santana-Morales, O., Escobedo-Olvera, M., Kacev, D., Castillo-Geniz,
- L., Graham, J. B., Rubin, R. D., Sosa-Nishizaki, O., & Rochin-Alamillo, A., (2013). The artisanal elasmobranch fishery of the Pacific coast of Baja California, Mexico. Fisheries Research, 108, 393-403.
- Randall, J. E. (1992). Review of the biology of the tiger shark (Galeocerdo cuvier). Australian Journal of Marine and Freshwater Research, 43(1), 21–31.
- Read, T. (2010). Mark-recapture of tiger shark (Galeocerdo cuvier) in New Caledonia: A photo-identification approach. Coral Reef Initiatives for the Pacific, 2-23.
- Reid, D. D., Robbins, W. D., & Peddemors, V. M. (2011, January). Decadal trends in shark catches and effort from the New South Wales, Australia, Shark Meshing Program 1950-2010. Marine and Freshwater Research, 62, 676-693.
- Simpfendorfer, C. (1992). Biology of tiger sharks (Galeocerdo cuvier) caught by the Queensland Shark Meshing Program off townsville, Australia. Australian Journal of Marine and Freshwater Research, 43, 3–43.
- Smith, S. E., Au, D. W., & Show, C. (1998). Intrinsic rebound potentials of 26 species of Pacific sharks. Marine and Freshwater Research, 49(7), 663-678.
- Sousa, B. P. (2012, August 3). Sharks Caught in Mozambican Waters. Cape Town, South Africa: 8th Working Party on Ecosystem and By Catch.
- Spaet, J. L. Y. & Berumen, M. L. (2015). Fish market surveys indicate unsustainable elasmobranch fisheries in the Saudi Arabian Red Sea. Fisheries Research, 161: 356–364.
- Spaet, J. L. Y, Nanninga, G. B., & Berumen, M. L. (2016). Ongoing decline of shark populations in the Eastern Red Sea. Biological Conservation, 201, 20-28.
- Stevens, J. D., Davis, T. L. O., & Church, A. G. (1982). NT shark gillnetting survey shows potential for Australian fishermen. Australian Fisheries, 41: 39–43.
- Tang, D. & Newbound, D. (2004). A new species of copepod (Siphonostomatoida: Caligidae) parasitic on the tiger shark Galeocerdo cuvier (Péron & Lesueur) from Western Australian waters. Systematic Parasitology, 58(1), 69-80.
- Thomson, K. S. & Simanek, D. E. (1977). Body form and locomotion in sharks. American Zoologist, 17(2), 343-354.
- Tillett, B. J., Field, I. C., Bradshaw, C. J. A., Johnson, G., Buckworth, R. C., Meekan, M. G., & Ovenden, J. R. (2012). Accuracy of species identification by fisheries observers in a north Australian shark fishery. Fisheries Research, 127, 109-115.
- Tobuni, I. M., Benabdallah, B. A. R., Serena, F., & Shakman, E. A. (2016). First documented presence of Galeocerdo cuvier (Péron & Lesueur, 1822) (Elasmbobranchii, Carcharhinidae) in the Mediterranean basin (Libyan waters). Marine Biodiversity Records, 91(1), 94.
- Tsehaye, I., Machiels, M. A. M., & Nagelkerke, L. A. J. (2007). Rapid shifts in catch composition in the artisanal Red Sea reef fisheries of Eritrea. Fisheries Research, 86, 58–68.
- Valinassab, T., Daryanabard, R., Dehghani, R., & Pierce, G. J. (2006). Abundance of demersal fish resources in the Persian Gulf and Oman Sea. Journal of the Marine Biological Association of the United Kingdom, 86, 1455-1462.
- Ward-Paige, C. A., Mora, C., Lotze, H. K., Pattengill-Semmens, C., McClenachan, L., Arias-Castro, E., & Myers, R. A. (2010). Large-scale absence of sharks on reefs in the greater-Caribbean: A footprint of human pressures. Public Library of Science (Plos) One, 5, e11968.
- Werry, J. M., Planes, S., Berumen, M. L., Lee, K. A., Braun, C. D., & Clua, E. (2014). Reef-fidelity and migration of tiger sharks, Galeocerdo cuvier, across the Coral Sea. Public Library of Science (Plos) One 9, e83249.
- White, W. T. (2007). Aspects of the biology of carcharhiniform sharks in Indonesian waters. Journal of the Marine Biological Association of the United Kingdom, 87, 1269-1276.
- Whitney, N. M. & Crow, G. L. (2007). Reproductive biology of the tiger shark (Galeocerdo cuvier) in Hawaii. Marine Biology, 151, 63-70.
- The Wikimedia Foundation. (2020, January 28). Tiger shark. Wikipedia.
- Wirsing, A., Heithaus, M., & Dill, L. (2006). Tiger shark (Galeocerdo cuvier) abundance and growth in a subtropical embayment: Evidence from 7 years of standardized fishing effort. Marine Biology, 149(4), 961-968.